According to a recent DelveInsight report, the Urea Cycle Disorders (UCD) market is expected to witness substantial expansion through 2032, driven by increasing disease awareness, improved diagnostics, and a rapidly advancing therapeutic pipeline. The report, titled “Urea Cycle Disorders Market Insights, Epidemiology, and Market Forecast – 2034,” provides a comprehensive assessment of market trends, historical and forecasted epidemiology, and treatment dynamics across the United States, EU4 (Germany, France, Italy, Spain), the United Kingdom, and Japan.
DelveInsight’s analysis indicates that the UCD market is projected to grow at a notable CAGR during the 2020-2034 forecast period, supported by rising diagnosis rates and multiple late- and mid-stage pipeline therapies. Increasing focus on rare metabolic disorders and strong regulatory incentives are further accelerating innovation in this space.
Get a Free sample for the Urea Cycle Disorders Market Report:
https://www.delveinsight.com/report-store/urea-cycle-disorders-market
Key Urea Cycle Disorders Market Developments
Recent clinical and regulatory milestones are reshaping the UCD landscape. In March 2025, iECURE reported encouraging results from its OTC-HOPE Phase I/II trial, where the first infant treated with ECUR-506 achieved complete clinical response, demonstrating restored OTC enzyme activity. Earlier, in April 2024, the US FDA cleared iECURE’s IND application for ECUR-506 in OTC deficiency.
Meanwhile, CAMP4 Therapeutics initiated a Phase I trial of CMP-CPS-001, a first-in-class regulatory RNA therapy, while Zevra Therapeutics refined its commercialization strategy for OLPRUVA in collaboration with the National Urea Cycle Disorders Foundation to improve patient education and early ammonia testing. Additionally, the European Commission’s approval of LOARGYS (pegzilarginase) in late 2023 marked a major milestone as the first disease-modifying therapy for ARG1-D, with US resubmission planned.
For an in-depth understanding of the Urea Cycle Disorders market outlook, drug uptake, treatment landscape, and epidemiology trends, click here: Urea Cycle Disorders Market Forecast
Urea Cycle Disorders Epidemiology and Disease Burden
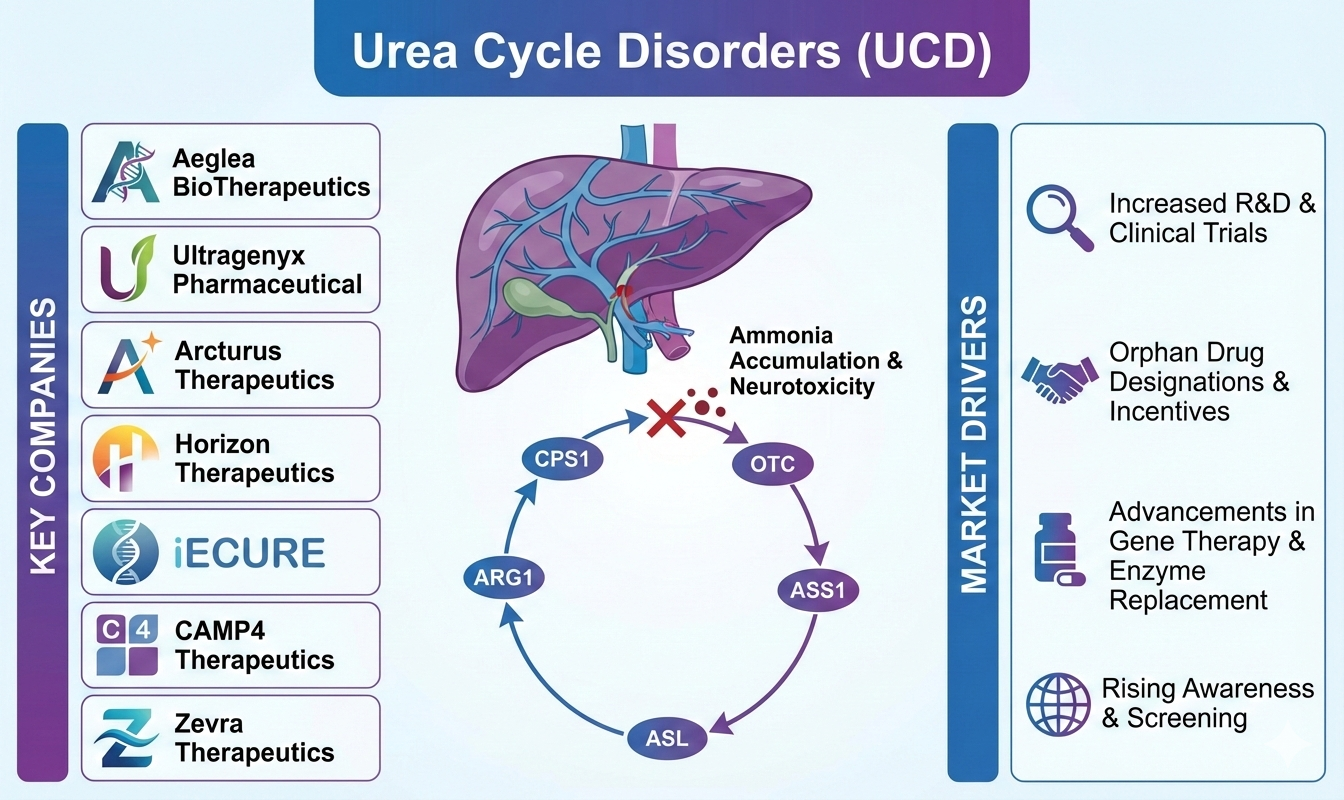
UCDs are rare genetic metabolic disorders characterized by impaired ammonia detoxification. According to CDC data, UCDs occur in approximately 1 in 8,500 births, though many cases remain undiagnosed. Studies indicate that OTC deficiency accounts for nearly two-thirds of cases, disproportionately affecting males, often with symptom onset in early infancy.
Urea Cycle Disorders Epidemiology Segmentation:
The Urea Cycle Disorders market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Urea Cycle Disorders Type-Specific Diagnosed Prevalent Cases
- Urea Cycle Disorders Onset-Specific Diagnosed Prevalent Cases
- Total Urea Cycle Disorders Diagnosed Prevalent Cases
- Urea Cycle Disorders Onset-specific Diagnosed Prevalent Cases
- Urea Cycle Disorders Treated Cases
Download the report to learn about the key factors influencing Urea Cycle Disorders epidemiology trends at the Urea Cycle Disorders Epidemiology Forecast
Urea Cycle Disorders Competitive Landscape and Pipeline
The Urea Cycle Disorders (UCD) competitive landscape features a dynamic mix of biopharmaceutical innovators, each advancing therapies that target key enzyme deficiencies responsible for UCDs. Traditional management focused on dietary restriction and ammonia-lowering medications like RAVICTI is being reshaped by emerging disease-modifying therapies, novel biologics, gene therapies, and metabolic modulators.
Key Urea Cycle Disorders Companies In The Treatment Landscape
Leading companies actively developing UCD treatments include Aeglea BioTherapeutics (NASDAQ: SYRE), Ultragenyx Pharmaceutical (NASDAQ: RARE), Arcturus Therapeutics (NASDAQ: ARCT), Horizon Therapeutics (NASDAQ: HZNP), iECURE, CAMP4 Therapeutics (NASDAQ: CAMP), Zevra Therapeutics (NASDAQ: ZVRA), and others. Established firms such as Horizon continue to commercialize existing therapies, while newer entrants focus on next-generation assets.
Urea Cycle Disorders Pipeline Highlights
The UCD pipeline is expanding rapidly with candidates in various stages of clinical development:
- ECUR-506 (iECURE) – Demonstrated promising Phase I/II results in OTC deficiency, showing enzyme activity restoration.
- CMP-CPS-001 (CAMP4 Therapeutics) – A first-in-class regulatory RNA therapy dosing its first patient in Phase I.
- ARCT-810 (Arcturus Therapeutics) – Gene therapy candidate targeting OTC deficiency.
- AEB1102 (pegzilarginase, Aeglea BioTherapeutics) – Approved in Europe for ARG1-D and planned for US resubmission.
- DTX301 (Ultragenyx) – Gene therapy for OTC deficiency with ongoing clinical evaluation.
- HPN-100 (Horizon) – Additional pipeline agent addressing metabolic pathways involved in UCD.
Urea Cycle Disorders Emerging Modalities
The pipeline increasingly includes gene therapy, mRNA therapy, regulatory RNA approaches, and improved enzyme replacement candidates. These modalities aim not only to manage symptoms but to correct underlying metabolic defects, reduce lifelong dependence on ammonia scavengers, and improve patient quality of life.
Urea Cycle Disorders Competitive Dynamics
Competition is shaped by innovation velocity, regulatory designations (e.g., orphan status), strategic collaborations, and clinical differentiation. Companies advancing therapies with superior safety profiles and durable efficacy are positioned to capture meaningful market share. DelveInsight’s market research provides comprehensive analysis of competitive positioning, pipeline progress, and strategic landscape evolution.
Urea Cycle Disorders Therapies and Key Companies
- DTX301: Ultragenyx Pharmaceutical
- LUNAR-OTC (ARCT-810): Arcturus Therapeutics
- ECUR-506: iECURE
- AEB1102 (pegzilarginase): Aeglea BioTherapeutics
- DTX301: Ultragenyx Pharmaceutical
- ARCT-810: Arcturus Therapeutics
- HPN-100: Horizon Pharma
- RAVICTI: Horizon Therapeutics, LLC
To learn more about companies active in the Urea Cycle Disorders treatment market, visit Urea Cycle Disorders Clinical Trials and Therapeutic Assessment
Conclusion:
With expanding pipelines, increasing awareness, and innovation in disease-modifying therapies, the Urea Cycle Disorders market is set for meaningful growth. DelveInsight’s in-depth market intelligence continues to support stakeholders with actionable insights into epidemiology trends, drug uptake, and competitive positioning.
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.