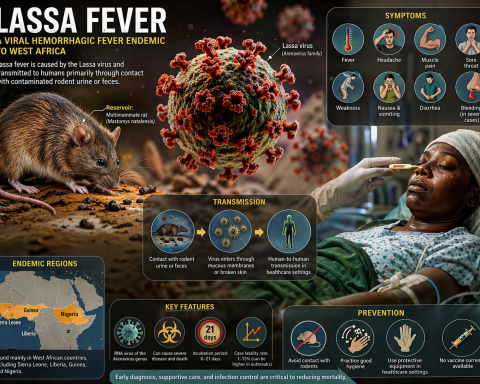
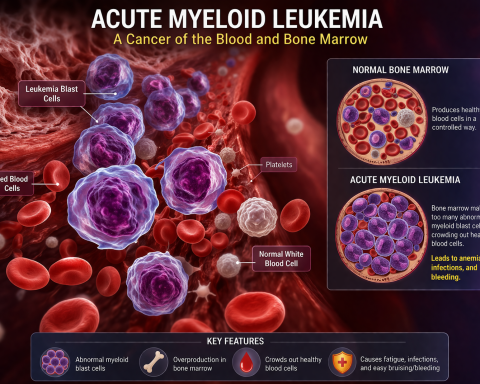
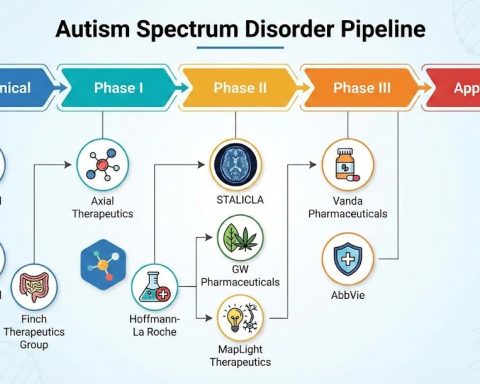
Global AI in Medical Diagnostics Market Set for Exceptional Growth at a CAGR of ~22% by 2032 | DelveInsight
The global artificial intelligence (AI) in medical diagnostics market is expanding at a rapid pace, driven by the rising incidence of chronic, genetic, and infectious diseases. AI-powered diagnostic solutions enable earlier detection, more accurate diagnoses, and personalized treatment planning by analyzing complex