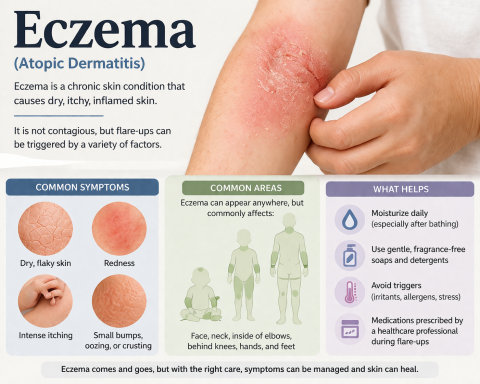
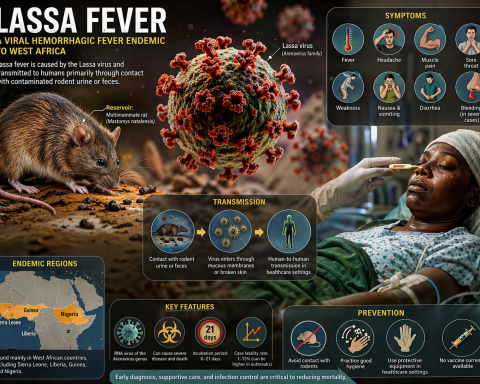
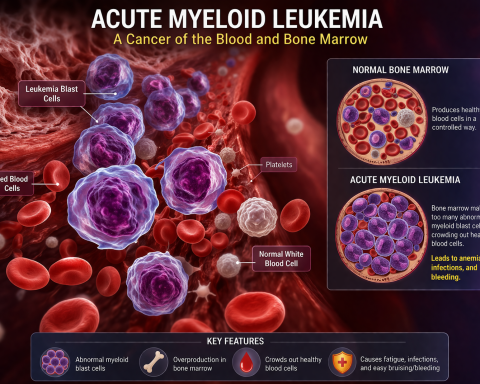
Inflammatory Myositis Market Set for Remarkable Growth: Projected CAGR of 11.7% Through 2034 Driven by Emerging Therapies and Rising Patient Demand Across Seven Major Markets
Inflammatory Myositis Market In 2023, the Inflammatory myositis market in the US stood out prominently among the seven major markets (7MM), reaching an impressive USD 301 million. This significant figure not only underscores the current robust demand but also highlights the substantial