Multiple Sclerosis Pipeline 2025: Clinical Progress, MOA, ROA, and Regulatory Advances – DelveInsight
Las Vegas, Nevada, United States – DelveInsight has released its latest industry report, “Multiple Sclerosis Pipeline Insight, 2025,” delivering a comprehensive overview of the evolving Multiple Sclerosis (Multiple Sclerosis) therapeutic landscape. According to DelveInsight’s analysis, more than 60 global pharmaceutical and biotechnology companies are actively developing 75+ Multiple Sclerosis therapies, spanning early discovery through late-stage clinical development and commercialization.
The report provides an in-depth evaluation of pipeline candidates, highlighting mechanisms of action (MOA), routes of administration (ROA), clinical trial progress, regulatory milestones, and competitive dynamics. It covers therapies across preclinical, Phase I, Phase II, Phase III, and marketed stages, offering stakeholders a holistic view of innovation trends shaping the Multiple Sclerosis market.
Get a Free Sample PDF Report to know more about Multiple Sclerosis Pipeline Therapeutic Assessment:
https://www.delveinsight.com/report-store/multiple-sclerosis-competitive-landscape
Expanding Multiple Sclerosis Clinical and Commercial Landscape
DelveInsight’s Multiple Sclerosis pipeline assessment details each drug’s scientific rationale, clinical data, development status, NDA approvals (where applicable), and strategic activities such as collaborations, licensing deals, mergers and acquisitions, funding, and regulatory designations. The report underscores the growing shift toward targeted immunology, neuroprotection, remyelination, and B-cell and BTK inhibition strategies.
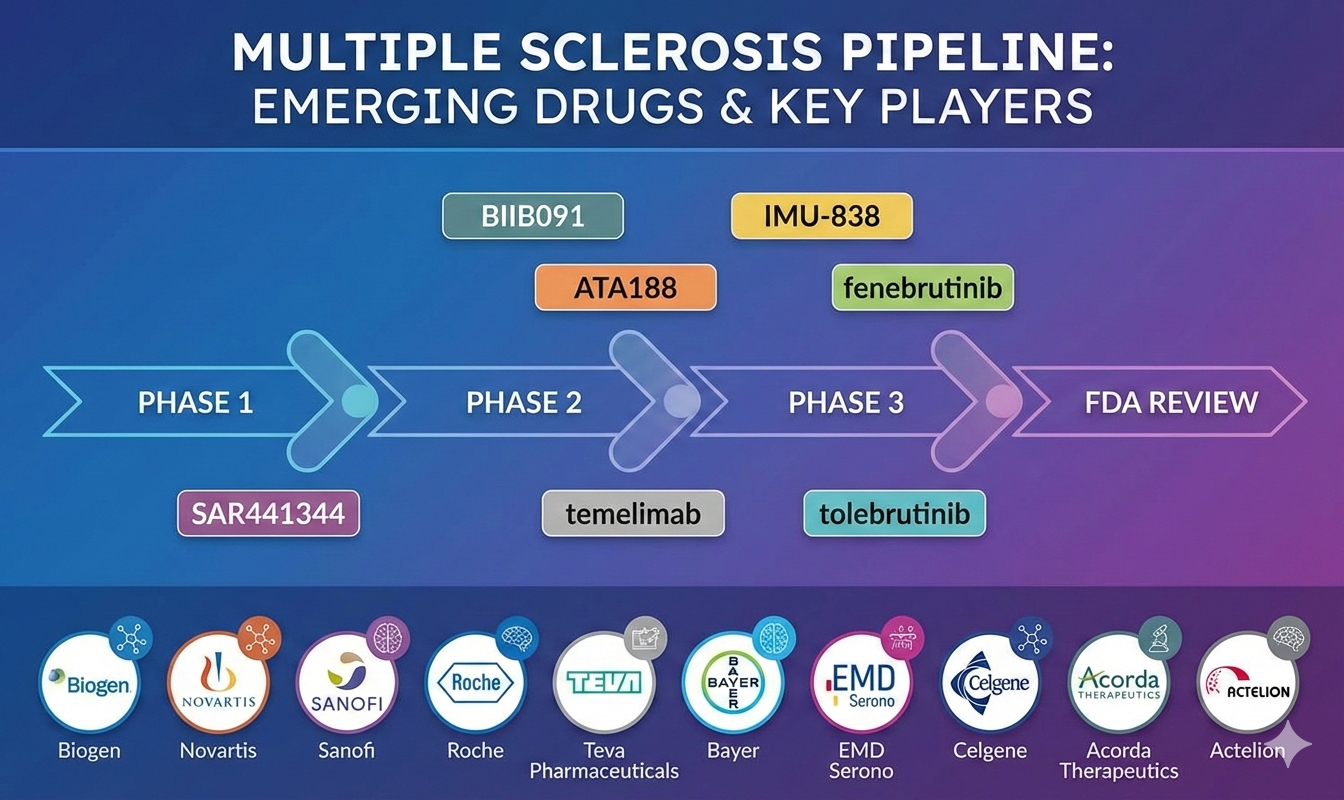
Key companies driving innovation include Biogen, Novartis, Sanofi, Roche, Teva Pharmaceuticals, Bayer, EMD Serono, Celgene, Acorda Therapeutics, and Actelion, along with emerging biotech players worldwide.
Multiple Sclerosis Overview
Multiple Sclerosis is a chronic autoimmune disease of the central nervous system characterized by inflammation, demyelination, and neurodegeneration. While no cure currently exists, advancements in disease-modifying therapies continue to improve symptom management and slow disease progression.
Recent Developments In the Multiple Sclerosis Treatment Landscape:
- In December 2025, The U.S. Food and Drug Administration (FDA) has granted tentative approval to Lupin’s Siponimod tablets, a bioequivalent version of Mayzent (siponimod), for the treatment of adults with relapsing forms of multiple sclerosis (Multiple Sclerosis). The therapy is approved for patients with clinically isolated syndrome, relapsing-remitting Multiple Sclerosis (RRMultiple Sclerosis), or active secondary progressive Multiple Sclerosis (SPMultiple Sclerosis). Like Novartis’ Mayzent, Lupin’s generic will be available in three strengths: 0.25 mg, 1 mg, and 2 mg tablets.
- In December 2025, The introduction of natalizumab-sztn (Tyruko) represented a significant milestone for biosimilars in neurology, becoming the first biosimilar approved for multiple sclerosis (Multiple Sclerosis) and the first neurology biosimilar to gain regulatory clearance in the U.S. Developed by Polpharma Biologics and marketed by Sandoz, Tyruko offers the potential to increase patient access to a high-efficacy biologic therapy for Multiple Sclerosis while fostering long-awaited price competition in this costly treatment area.
- In November 2025, Zydus Lifesciences’ multiple sclerosis therapy, Diroximel Fumarate, has received final approval from the U.S. FDA. The drug is indicated for the treatment of relapsing forms of multiple sclerosis in adults and holds an estimated market value of approximately USD 9,994 million (~INR 8,500 crore) in the United States.
- In June 2025, Ashvattha Therapeutics announced at the SNMMI Annual Meeting that its nanomedicines can be tailored to visualize neuroinflammation in patients with Multiple Sclerosis.
- In June 2025, IASO Bio showcased encouraging results of Equecabtagene Autoleucel in Multiple Sclerosis patients from an IIT study during the 11th Congress of the EAN.
- In June 2025, Telomir Pharmaceuticals announced that Telomir-1 resets the body’s epigenetic clock, reverses DNA methylation, and restores gene regulation in patients with Werner Syndrome.
- In May 2025, TG Therapeutics announced upcoming data presentations on BRIUMVI for Multiple Sclerosis at the 2025 Consortium of Multiple Sclerosis Centers (CMultiple SclerosisC) Annual Meeting.
- In May 2025, Immunic announced plans to proceed with the development of its multiple sclerosis (Multiple Sclerosis) candidate, vidofludimus calcium (IMU-838), despite its Phase II trial not meeting the primary endpoint. Preliminary results from the ongoing CALLIPER study (NCT05054140) revealed a modest advantage in an exploratory MRI-based primary endpoint, showing a 5% annualized reduction in brain volume loss compared to placebo.
- In March 2025, The U.S. FDA has granted Priority Review to Sanofi’s regulatory submission for tolebrutinib, following positive outcomes from the Phase III HERCULES (NCT04411641), GEMINI 1 (NCT04410978), and GEMINI 2 (NCT04410991) trials. The submission seeks approval for the treatment of non-relapsing secondary progressive multiple sclerosis (nrSPMultiple Sclerosis) and aims to reduce disability progression unrelated to relapse activity in adults.
- In February 2025, Quantum BioPharma Ltd. (NASDAQ: QNTM) (CSE: QNTM) (FRA: 0K91) announced today the completion of its trial titled “A Phase 1, Randomized, Double-Blind, Placebo-Controlled, Multiple Ascending Dose Study to Evaluate the Safety and Pharmacokinetics of Lucid-21-302 in Healthy Adult Participants.” Following the trial, a final safety review committee (SRC) meeting concluded that Lucid-21-302 (Lucid-Multiple Sclerosis) was well-tolerated, with no safety concerns and no serious adverse events reported. Lucid-Multiple Sclerosis is a first-in-class, non-immunomodulatory, neuroprotective compound being developed for the treatment of multiple sclerosis (Multiple Sclerosis)
- In January 2025, Pheno Therapeutics received clinical trial authorization (CTA) from the UK’s Medicines and Healthcare Products Regulatory Agency (MHRA) for its small-molecule therapy, PTD802. This approval paves the way for the first-in-human trial of PTD802, a selective G protein-coupled receptor 17 (GPR17) antagonist, designed to promote remyelination in patients with neurological disorders, particularly multiple sclerosis (Multiple Sclerosis).
- In January 2025, Contineum Therapeutics, Inc. (NASDAQ: CTNM), a clinical-stage biopharmaceutical company specializing in innovative therapies for neuroscience, inflammation, and immunology (NI&I) conditions, announced the successful completion of targeted enrollment for its Phase 2 PIPE-307 VISTA trial. A total of 168 patients have been enrolled in the study, which is evaluating PIPE-307, a potentially first-in-class M1 receptor antagonist, as a treatment for relapsing-remitting multiple sclerosis (RRMultiple Sclerosis).
- In September 2024, Roche presented additional data from a Phase II study of its Bruton’s tyrosine kinase (BTK) inhibitor, revealing nearly complete disease suppression in patients with relapsing multiple sclerosis (RMultiple Sclerosis) based on long-term findings. The company shared 48-week data for fenebrutinib from the FENopta open-label extension (OLE) study (NCT05119569), which followed a 12-week double-blind Phase II trial. This update comes shortly after Sanofi reported mixed results for its BTK inhibitor, tolebrutinib, where the HERCULES trial met its primary endpoint, but the two GEMINI trials did not.
Further Multiple Sclerosisproduct details are provided in the report. Download the Multiple SclerosisCompetitive Landscape report to learn more about the emerging Multiple Sclerosis therapies
Emerging Multiple Sclerosis Drugs Under Different Phases of Clinical Development Include:
- BIIB091: Biogen
- NeuroVax: Immune Response BioPharma
- IMCY-0141: ImCyse
- ATA188: Atara Biotherapeutics
- EHP-101: Emerald Health Pharmaceuticals
- SAR441344: Sanofi
- RC18: RemeGen
- GB7208: Gossamer Bio
- MRx0002: 4D Pharma
- HuL001: HuniLife Biotechnology
- ANK-700: Anokion
- GSK3888130: GSK plc
- Temelimab: GeNeuro
- IMU-838: Immunic Therapeutics
- Tolebrutinib: Sanofi
- Fenebrutinib: Hoffman-La-Roche
Multiple Sclerosis Notable Pipeline and Regulatory Developments
Recent regulatory and clinical highlights shaping the Multiple Sclerosis pipeline include:
- FDA tentative approval of Lupin’s Siponimod tablets, a generic version of Mayzent, for relapsing forms of Multiple Sclerosis
- Approval of natalizumab-sztn (Tyruko), the first Multiple Sclerosis biosimilar and the first neurology biosimilar approved in the U.S.
- Final FDA approval of Zydus Lifesciences’ Diroximel Fumarate for relapsing Multiple Sclerosis
- Priority Review granted to Sanofi’s tolebrutinib for non-relapsing secondary progressive Multiple Sclerosis
- Encouraging clinical updates for BTK inhibitors, CAR-T therapies, stem cell approaches, and neuroprotective compounds
Emerging therapies such as BIIB091, ATA188, IMU-838, fenebrutinib, tolebrutinib, temelimab, and SAR441344 are expected to significantly influence the Multiple Sclerosis treatment paradigm over the coming years.
Download Sample PDF Report to know more about Multiple Sclerosisdrugs and therapies
Multiple Sclerosis Market Drivers
- Rising global prevalence of multiple sclerosis, particularly in North America and Europe
- Expanding aging population and improved life expectancy of Multiple Sclerosis patients
- Strong and diverse R&D pipeline with innovative disease-modifying therapies (DMTs)
- Increasing adoption of biologics, BTK inhibitors, CAR-T, and neuroprotective therapies
- Growing awareness and early diagnosis supported by advanced imaging and biomarkers
- Favorable regulatory support, including fast-track and priority review designations
- Entry of biosimilars and generics improving treatment accessibility
- Increased healthcare expenditure and reimbursement support in developed markets
Multiple Sclerosis Market Barriers
- High cost of branded disease-modifying therapies limiting patient access
- Risk of serious adverse events and long-term safety concerns
- Treatment adherence challenges due to chronic dosing and side effects
- Stringent regulatory requirements and lengthy clinical trial timelines
- Limited efficacy of existing therapies in progressive forms of Multiple Sclerosis
- Regional disparities in access to advanced Multiple Sclerosis care and diagnostics
- Pricing pressure and competition from biosimilars affecting revenue growth
Conclusion:
DelveInsight’s Multiple Sclerosis Pipeline Insight 2025 report offers a strategic and data-driven perspective on one of the most dynamic therapeutic areas in neurology. With strong innovation momentum and multiple late-stage candidates nearing regulatory decisions, the Multiple Sclerosis market is poised for meaningful transformation in the coming years.
Request a free sample PDF to explore the complete Multiple Sclerosis pipeline analysis and emerging therapy insights
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.