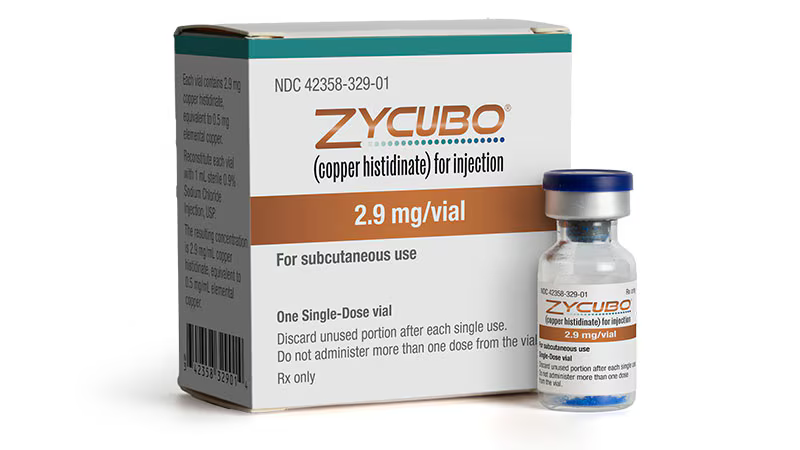
Sentynl Therapeutics, a US-based biopharmaceutical subsidiary of Zydus Lifesciences Limited, has achieved a landmark regulatory milestone with FDA approval of ZYCUBO (copper histidinate) injection for the treatment of pediatric patients with Menkes disease. This marks the first and only FDA-approved therapy for a condition that has claimed countless young lives in the absence of any effective treatment option.
The Menkes Disease treatment landscape is evolving rapidly. You can learn more @ https://www.delveinsight.com/report-store/menkes-disease-market
Menkes Disease: A High Unmet Need
Menkes disease,a rare, X-linked recessive genetic disorder affecting copper metabolism, has represented one of pediatric medicine’s most heartbreaking dilemmas. The condition stems from mutations in the ATP7A gene, which encodes a copper transporter essential for cellular copper uptake. Without functional copper transport, affected infants experience catastrophic neurological deterioration, characterized by seizures, severe developmental delays, hypotonia, and distinctive kinky hair. Historically, untreated patients rarely survived beyond age three.
The disease affects an estimated 1 in 8,664 to 1 in 35,000 live male births, rendering it a rare but devastating diagnosis that offers families no approved therapeutic recourse—until now.
“Approval is a pivotal step towards achieving our goal of making a meaningful impact on patients, caregivers, and the rare disease community,” said Dr. Sharvil P. Patel, Managing Director of Zydus Lifesciences Limited. “This milestone marks a transformative moment for the Zydus Group and for families affected by Menkes disease. For the first time, patients have access to an approved therapy, offering hope where no options existed.”
ZYCUBO Clinical Evidence Demonstrates Transformative Efficacy
ZYCUBO’s approval is grounded in compelling clinical evidence from two open-label, single-arm trials encompassing 129 pediatric patients treated with copper histidinate—among the largest prospective cohorts assembled for this ultra-rare indication. The data reveal remarkable survival improvements that fundamentally reshape the natural history of Menkes disease.
Early treatment outcomes: Patients who received ZYCUBO within the first four weeks of life experienced a 79% reduction in mortality risk compared to untreated historical controls. Median overall survival surged to 177.1 months versus 16.1 months in the control cohort—a transformation translating to approximately 14.8 years versus roughly 1.3 years. Notably, nearly 50% of early-treated patients survived beyond six years, while untreated controls achieved no survival extending beyond six years.
Late treatment outcomes: Even patients initiating therapy after the critical early window demonstrated significant benefit. Late-treated cohorts showed a 75% reduction in mortality risk with median overall survival of 62.4 months compared to 17.6 months in untreated controls.
Beyond these survival metrics, clinical evaluations revealed functional improvements that extend quality of life considerably. Patients completing three years of early treatment were able to attend school, remain cognitively engaged, and participate in age-appropriate activities—outcomes inconceivable in the pre-treatment era.
“This milestone represents the culmination of decades of research into better understanding and ultimately finding an effective treatment for Menkes disease,” said Dr. Stephen Kaler, a clinical genetics and genomics specialist at Columbia University Medical Center. “Increased awareness of Menkes disease and rapid testing upon suspicion are critical, as beginning copper histidinate therapy in affected neonates has been shown to reduce symptoms and prolong life.”
ZYCUBO Mechanism and Clinical Administration
ZYCUBO operates through a targeted biochemical strategy: the subcutaneous injectable copper histidinate formulation delivers bioavailable elemental copper directly into the systemic circulation, effectively bypassing the genetic defect that impairs intestinal copper absorption. By restoring copper homeostasis and maintaining physiologic copper levels across the blood-brain barrier—the critical site of neurological vulnerability in Menkes disease—the therapy addresses the fundamental pathophysiology underlying disease progression.
Clinical administration involves daily subcutaneous injection of 1,450 μg (equivalent to 250 μg elemental copper) twice daily until 12 months of age, then once daily thereafter, for a total treatment duration of up to three years. This regimen was specifically designed to deliver consistent copper replacement during the critical developmental period when neurological damage accrues most rapidly.
Copper Histidinate Safety Profile and Clinical Considerations
As with any therapy for pediatric patients with severe, multi-system genetic disorders, ZYCUBO carries important safety considerations that require vigilant medical monitoring. The most common adverse reactions reported (incidence ≥7%) include pneumonia (30%), viral infection (27%), respiratory failure (23%), seizure (23%), bacterial infection (20%), and hemorrhage (18%). Additional reactions include hypotension, vomiting, tachycardia, pyrexia, volume depletion, fracture, dyspnea, elevated transaminases, diarrhea, fungal infection, anemia, and local injection site reactions.
Significantly, the clinical trial data revealed that no patients discontinued treatment due to adverse events considered related to the study drug—a finding underscoring that observed complications largely reflect underlying disease severity rather than drug-induced toxicity.
However, the prescribing information includes important warnings regarding potential copper accumulation and toxicity, particularly affecting the kidneys, liver, and hematopoietic system. Because Menkes disease patients already suffer copper accumulation from failed transporter function, clinicians must implement rigorous monitoring protocols for renal and hepatic function, as well as hematologic parameters, with dose adjustments guided by laboratory findings. This requirement underscores that ZYCUBO’s use demands specialist-level clinical management in experienced pediatric centers.
FDA Regulatory Pathway and Strategic Designations
The FDA’s approval reflects recognition of ZYCUBO’s unprecedented clinical importance. Sentynl received multiple expedited designations supporting development:
- Breakthrough Therapy Designation (recognizing substantial improvement over available alternatives)
- Fast Track Designation (enabling priority review)
- Orphan Drug Designation (reflecting ultra-rare indication status)
- Rare Pediatric Disease Designation (enabling priority review voucher eligibility)
Notably, Sentynl encountered a Complete Response Letter (CRL) from the FDA on September 30, 2025, citing manufacturing compliance observations rather than efficacy or safety concerns. Following expedited resubmission of a revised New Drug Application on November 14, 2025, the FDA issued approval within two months—reflecting the agency’s confidence in the underlying clinical data and the drug’s unmet need status.
European regulators have also granted Orphan Medicinal Product Designation to copper histidinate, signaling international recognition of the therapy’s importance.
A Strategic Milestone for Zydus Lifesciences
This approval represents a watershed moment for Zydus Lifesciences, traditionally known as a global generics powerhouse with commanding positions in cost-sensitive pharmaceutical markets. The ZYCUBO approval marks the company’s entry into innovation-led rare disease therapeutics—a strategic repositioning that signals management’s commitment to advancing high-impact specialty treatments for patient populations with devastating conditions.
Sentynl acquired the copper histidinate asset from Cyprium Therapeutics in 2023 for a transaction valued at over $134 million, including approximately $129 million in milestone payments and royalty components. This investment reflects Zydus’s recognition of the program’s clinical potential and strategic importance.
“FDA’s approval serves as compelling affirmation that a safe and effective therapy is now available for patients living with this devastating disease,” said Matt Heck, CEO of Sentynl. “Menkes disease presents significant challenges for patients and their families. With no known cure, most untreated patients do not survive beyond three years of age.”
The approval also signals broader ambitions. Sentynl is advancing additional rare disease assets through its development pipeline, including Zokinvy (lonafarnib) for Hutchinson-Gilford Progeria Syndrome—positioning the company as a meaningful player in the orphan drug ecosystem.
Clinical Practice Implications and Diagnostic Imperatives
ZYCUBO’s availability creates new clinical imperatives around early detection. The dramatic survival differential between patients treated within four weeks of life versus later treatment underscores that rapid diagnosis is essential to therapeutic efficacy. Menkes disease diagnosis historically has been challenging due to the nonspecific nature of neonatal presentation—affected infants often present with hypothermia, hypoglycemia, or prolonged jaundice before characteristic features such as kinky hair emerge.
Clinicians should maintain heightened suspicion for Menkes disease in male neonates presenting with unexplained neurologic dysfunction, seizures, or failure to thrive, particularly when accompanied by distinctive hair abnormalities. Definitive diagnosis requires ATP7A genetic testing and biochemical confirmation (low serum copper and ceruloplasmin levels), though optimal outcomes demand diagnostic velocity that many centers have not historically prioritized for such rare conditions.
The approval may catalyze improvements in diagnostic infrastructure and awareness among neonatologists, genetic specialists, and pediatric neurologists—accelerating the time to diagnosis and treatment initiation.
Menkes Disease Market and Public Health Context
ZYCUBO’s approval arrives within a rapidly expanding orphan drug landscape. The global orphan drug market was valued at approximately $165 billion in 2023 and is projected to exceed $340 billion by 2030, reflecting a compound annual growth rate exceeding 10%. Notably, approximately 95% of rare diseases currently lack any approved therapeutic option—creating a vast frontier of unmet medical need.
Pediatric orphan drugs represent a particularly compelling segment, addressing diseases that predominantly affect children and carrying high humanitarian value. The FDA’s Rare Pediatric Disease Priority Review Voucher program and international orphan drug incentive frameworks have substantially catalyzed development of therapies for ultra-rare conditions, with ZYCUBO exemplifying this regulatory momentum.
Menkes Disease Unmet Need and Patient Impact
For families affected by Menkes disease, ZYCUBO represents the end of a diagnostic and therapeutic odyssey. Prior to approval, the “diagnostic odyssey” for Menkes disease often extended weeks or months—a period during which irreversible neurological damage accrued in affected infants. Parents faced the devastation of watching their children’s development plateau and regress, with counseling limited to supportive care and palliative management.
The ZYCUBO approval fundamentally reshapes the trajectory for newly diagnosed patients and creates opportunities for improved outcomes in the immediate perinatal period. Early diagnosis and treatment now translate to quantifiable survival and functional improvements—a shift from inevitable mortality to a condition manageable through specialized therapy.
Looking Forward
Sentynl’s regulatory success with ZYCUBO establishes a proof-of-concept for rare genetic metabolic disorders and positions the company to advance additional innovation-led therapies targeting ultra-rare pediatric indications. The approval also underscores Zydus Lifesciences’ strategic evolution beyond generic pharmaceuticals into innovation-led specialty care.
From a public health perspective, ZYCUBO’s availability underscores the critical importance of rapid diagnostic testing, clinical awareness among neonatologists, and access to specialized pediatric centers capable of managing the medical complexity inherent in copper replacement therapy. Equitable access and global availability remain important considerations for a condition affecting families across diverse socioeconomic strata.
The approval of ZYCUBO represents one of pediatric medicine’s singular achievements: the transformation of a fatal genetic disorder into a manageable, treatable condition—offering families affected by Menkes disease genuine hope and the prospect of a dramatically extended and more fulfilling life trajectory for their children.