According to DelveInsight’s latest report, “Dermatomyositis Market Insights, Epidemiology, and Market Forecast – 2034,” the global dermatomyositis market is expected to experience robust growth through 2034, driven by rising disease awareness, expanding diagnostic rates, and a rapidly advancing therapeutic pipeline. The report delivers comprehensive insights into market size, epidemiology trends, treatment landscape, and drug uptake across the United States, EU4 (Germany, France, Italy, Spain), the United Kingdom, and Japan.
Dermatomyositis Market Highlights
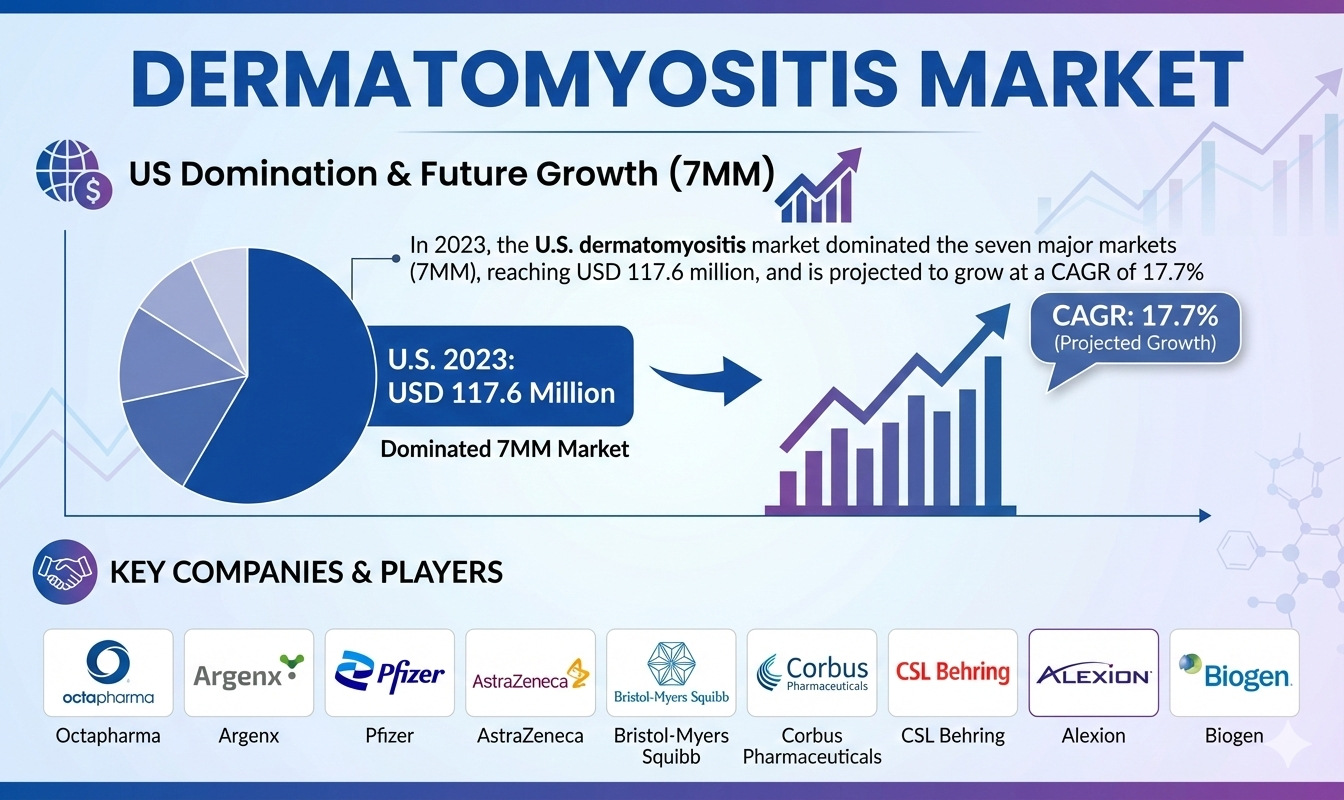
- In 2023, the U.S. dermatomyositis market dominated the seven major markets (7MM), reaching USD 117.6 million, and is projected to grow at a CAGR of 17.7% through 2034.
- The United States accounted for nearly 62% of the total 7MM market, surpassing Europe and Japan combined.
- Germany led the EU4 countries with a market size of USD 11.29 million, while Japan represented approximately USD 25.28 million, contributing 14% of the total market.
Dermatomyositis Overview
Dermatomyositis is a rare autoimmune inflammatory disease that primarily affects the muscles and skin. It is characterized by progressive muscle weakness, especially in the shoulders, hips, and neck, along with distinctive skin rashes such as reddish-violet discoloration on the face, knuckles, elbows, and knees. The condition occurs when the immune system mistakenly attacks healthy muscle and skin tissues. Dermatomyositis can also involve other organs, including the lungs, heart, and gastrointestinal tract, and may be associated with an increased risk of certain cancers. Early diagnosis and timely treatment are crucial to control symptoms, prevent complications, and improve quality of life.
Get a Free sample for the Dermatomyositis Market Report: https://www.delveinsight.com/report-store/dermatomyositis-market
Dermatomyositis Clinical and Regulatory Developments
The dermatomyositis treatment landscape is evolving rapidly with several late-stage and regulatory milestones:
- In September 2025, Roivant and its subsidiary Priovant announced that their investigational oral therapy for inflammatory diseases achieved success in a Phase 3 trial for the rare condition dermatomyositis, demonstrating a significant reduction in signs and symptoms compared to placebo over one year of treatment.
- In August 2025, Noxopharm Limited announced the successful completion of the second dose cohort of SOF-SKN™ in the HERACLES trial. The safety steering committee confirmed that this dose level is safe and well-tolerated, with no clinically significant concerns. The trial will now advance to the third cohort, receiving a higher dose. HERACLES is a first-in-human study of SOF-SKN™, a novel drug candidate targeting autoimmune diseases. The trial evaluates the safety and tolerability of four different dose levels and is conducted in Australia to leverage local expertise in lupus research and early-phase clinical trials, while also benefiting from federal R&D tax incentives. SOF-SKN is initially being developed for autoimmune conditions such as cutaneous lupus erythematosus (CLE), with potential expansion to other autoimmune-related skin diseases, including psoriasis and dermatomyositis.
- In February 2025, NovelMed Therapeutics, a clinical-stage biotech company focused on complement-targeting therapies, announced that the U.S. Food and Drug Administration (FDA) has approved Ruxoprubart (NM8074), an investigational drug, to commence a Phase II efficacy trial for the treatment of Dermatomyositis (DM), a rare autoimmune disease.
- In January 2025, RESTEM announced that the FDA has granted Fast Track designation to its Restem-L program, which utilizes umbilical cord outer lining stem cells (ULSCs) to treat Polymyositis and Dermatomyositis (PM/DM), now categorized under Idiopathic Inflammatory Myopathy (IIM). This development comes shortly after Restem-L received Orphan Drug Designation for IIM.
- In December 2024, RESTEM, a clinical-stage biotechnology company focused on developing off-the-shelf, next-generation cell therapies to modulate the immune system, announced that the U.S. Food and Drug Administration (FDA) has granted Orphan Drug Designation (ODD) to its umbilical cord outer lining stem cells (ULSCs) program for the treatment of Polymyositis (PM) and Dermatomyositis (DM).
- In July 2024, Priovant Therapeutics announced that it had completed enrollment for the Phase 3 VALOR study assessing brepocitinib in dermatomyositis. This study has included 241 participants from 90 locations across four continents, marking it as the largest interventional trial for dermatomyositis ever undertaken.
Dermatomyositis Epidemiology Insights
- In 2023, approximately 38,500 diagnosed dermatomyositis cases were reported in the United States.
- Women were disproportionately affected, with a 2:1 female-to-male ratio.
- The highest prevalence was observed in individuals aged 40–79 years.
- Germany recorded the highest number of cases among EU4 countries, while Spain reported the lowest.
Dermatomyositis Epidemiology Segmentation
The Dermatomyositis market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Prevalence of Dermatomyositis
- Prevalent Cases of Dermatomyositis by severity
- Gender-specific Prevalence of Dermatomyositis
- Diagnosed Cases of Episodic and Chronic Dermatomyositis
Download the report to understand which factors are driving Dermatomyositis epidemiology trends @ Dermatomyositis Epidemiology Forecast
Dermatomyositis Treatment Landscape and Market Dynamics
Current dermatomyositis management relies on corticosteroids, immunosuppressants, immunoglobulins, and biologics, with immunoglobulin therapies generating the highest revenue across the 7MM in 2023. Key companies shaping the market include Octapharma, Argenx, Pfizer, AstraZeneca, Bristol-Myers Squibb, Corbus Pharmaceuticals, CSL Behring, Alexion, Biogen, and Priovant.
Dermatomyositis Therapies and Key Companies
- OCTAGAM 10% (immunoglobulin): Octapharma
- Efgartigimod: Argenx
- Brepocitinib: Priovant Therapeutics, Inc.
- Lenabasum: Corbus Pharmaceuticals
- HIZENTRA: CSL Behring
- Ravulizumab: Alexion
- Lenabasum: Corbus Pharmaceuticals Inc.
- IMO-8400: Idera Pharmaceuticals
- GLPG3667: Galapagos NV
- M5049: EMD Serono
- human immunoglobulin G: CSL Behring
- PF-06823859: Pfizer
- Octagam: Octapharma
- KZR-616: Kezar Life Sciences, Inc.
- GB-0998: Japan Blood Products Organization
- H.P. Acthar Gel: Mallinckrodt
- Adrenocorticotropic Hormone Gel: Mallinckrodt
- tocilizumab: Genentech, Inc.
- Tacrolimus: Astellas Pharma Inc
- Rituximab: Biogen
Dermatomyositis Market Outlook
The dermatomyositis market outlook remains highly promising due to increasing prevalence, strong late-stage pipelines, and innovation across FcRn inhibitors, complement inhibitors, JAK inhibitors, and cell therapies. As novel disease-modifying treatments approach commercialization, market dynamics are expected to shift significantly during the forecast period.
Dermatomyositis Market Drivers
- Rising disease awareness and improved diagnosis due to better clinical guidelines and advanced diagnostic tools.
- Increasing prevalence of autoimmune and inflammatory disorders, supporting market expansion globally.
- Strong pipeline of emerging therapies, including biologics, FcRn inhibitors, JAK inhibitors, and complement-targeted drugs.
- Regulatory incentives such as Orphan Drug Designation, Fast Track, and Breakthrough Therapy status accelerating drug development.
- Growing unmet medical need, as current treatments are largely symptomatic and not disease-modifying.
- Expansion of clinical trials and R&D investments by pharmaceutical and biotech companies.
- Improved reimbursement frameworks in developed markets, enhancing patient access to advanced therapies.
Dermatomyositis Market Barriers
- Rarity of the disease, resulting in a small patient pool and limited commercial scalability.
- High cost of advanced biologics and immunotherapies, restricting access in price-sensitive regions.
- Lack of curative treatments, with most therapies focused on symptom management.
- Safety concerns and long-term side effects associated with corticosteroids and immunosuppressants.
- Diagnostic complexity and disease heterogeneity, leading to delayed or misdiagnosis.
- Limited clinical trial populations, which can slow regulatory approvals and market entry.
- Reimbursement challenges in emerging markets due to budget constraints and limited healthcare infrastructure.
To know more about Dermatomyositis companies working in the treatment market, visit @ Dermatomyositis Clinical Trials and Therapeutic Assessment
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.