The chemotherapy-induced peripheral neuropathy (CIPN) treatment market is emerging as a high-growth, high-value opportunity, fueled by a profound lack of approved therapies and a substantial unmet clinical need. As the global use of chemotherapy particularly neurotoxic agents continues to rise, the number of patients affected by CIPN is expanding steadily.
This growing patient pool, combined with the anticipated launch of several late-stage pipeline therapies such as Halneuron (Wex Pharmaceuticals), ATX01 (AlgoTx), ART-123 (Asahi Kasei Pharma/Veloxis Pharmaceuticals), WST-057 (WinSanTor, Inc.), and others, is expected to significantly accelerate market growth over the forecast period.
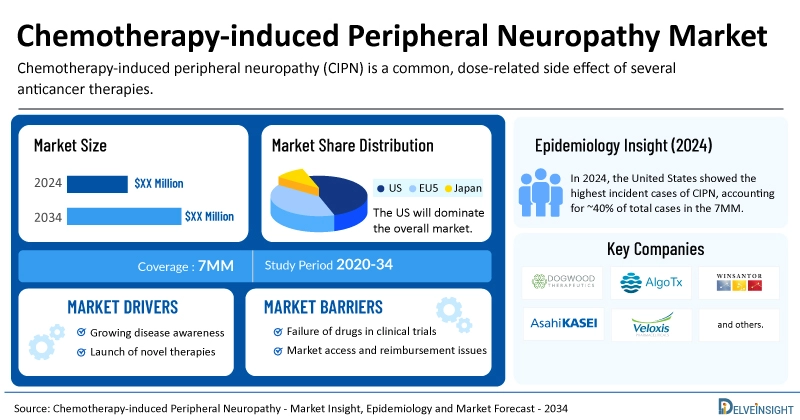
According to DelveInsight’s Chemotherapy-Induced Peripheral Neuropathy Market Insights report, the study provides a comprehensive assessment of current treatment practices, emerging CIPN therapies, competitive market share, and historical and forecasted market size from 2020 to 2034 across key geographies, including the United States, EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
👉 Explore which CIPN therapies are best positioned to capture market share in the coming decade: https://www.delveinsight.com/sample-request/cipn-market
Chemotherapy-Induced Peripheral Neuropathy Market Overview
- The total CIPN treatment market is projected to experience sustained growth across major markets through 2034.
- The United States represents the largest share of the global CIPN market, surpassing EU4, the UK, and Japan.
- In 2024, the U.S. accounted for approximately 40% of total CIPN incident cases across the 7MM, with prevalence expected to rise at a steady CAGR through 2034.
- Leading companies such as Dogwood Therapeutics, AlgoTx, Asahi Kasei Pharma, Veloxis Pharmaceuticals, and WinSanTor, Inc. are actively advancing innovative CIPN therapies.
- Several late-stage candidates including Halneuron (tetrodotoxin), ATX01, ART-123 (recombinant thrombomodulin alfa), and WST-057 are anticipated to enter the market during the forecast period, potentially reshaping the treatment landscape.
Key Drivers Fueling Chemotherapy-Induced Peripheral Neuropathy Market Growth
Rising Cancer Incidence and Chemotherapy Utilization
The global increase in cancer prevalence driven by aging populations, lifestyle changes, and environmental factors has led to broader chemotherapy use. Improved cancer survival rates have resulted in prolonged exposure to neurotoxic agents, increasing both the incidence and awareness of CIPN.
Growing Acceptance of Multimodal and Non-Pharmacological Approaches
Supportive care strategies such as acupuncture, transcutaneous electrical nerve stimulation (TENS), and other integrative therapies are gaining traction as adjuncts to symptom management, enhancing quality of life for affected patients.
Advancement of Novel CIPN Therapies
Several first- and best-in-class therapies are progressing through late-stage development, offering the potential for more targeted and effective management of CIPN compared to current off-label options.
👉 Gain deeper insight into the clinical and commercial drivers transforming the CIPN market
CIPN Market Landscape and Treatment Paradigm
Currently, no therapies are specifically approved for the treatment or prevention of CIPN, and management relies heavily on off-label use of medications for neuropathic pain. These include alpha-2-delta ligands, SNRIs, tricyclic antidepressants, opioids, and topical analgesics, which provide symptomatic relief but lack disease-modifying efficacy.
Late-stage investigational therapies such as Halneuron, ATX01, and ART-123 are expected to enter the market between 2025 and 2034, potentially driving a paradigm shift toward more targeted and mechanism-based interventions across the 7MM.
Chemotherapy-Induced Peripheral Neuropathy Competitive Landscape Highlights
- Halneuron (Tetrodotoxin, TTX) by Wex Pharmaceuticals is a small-molecule therapy designed to treat moderate-to-severe neuropathic pain by blocking voltage-gated sodium channels. The drug has received FDA Fast Track designation for chemotherapy-induced neuropathic pain and is currently in a Phase IIb clinical trial.
- ATX01 (AlgoTx) is a first-in-class topical therapy formulated to act locally within damaged nerve fibers, minimizing systemic exposure. The drug has successfully completed the Phase II ACT study in the U.S. and Europe.
- ART-123 (Asahi Kasei Pharma/Veloxis Pharmaceuticals) is a recombinant thrombomodulin with anticoagulant and anti-inflammatory properties. It is currently undergoing Phase III evaluation in the United States for CIPN treatment.
The successful approval and commercialization of these therapies are expected to transform the CIPN treatment landscape, establishing new standards of care and unlocking meaningful commercial opportunities.
👉Explore innovation trends shaping next-generation CIPN therapies
Recent Developments in the CIPN Market
- August 2025: Dogwood Therapeutics enrolled the first 50 patients in its Phase IIb CINP trial and remains on track for an interim analysis later in the year.
- June 2025: Asahi Kasei Pharma initiated dosing in a Phase III study of ART-123 in Japan for the prevention of CIPN-related sensory symptoms.
- February 2025: AlgoTherapeutix completed enrollment in its 276-patient Phase II ACT trial evaluating ATX01 in CIPN.
Key CIPN Market Companies
Leading Chemotherapy-Induced Peripheral Neuropathy companies such as
- Dogwood Therapeutics
- AlgoTx
- Asahi Kasei Pharma
- Veloxis Pharmaceuticals
- and WinSanTor, Inc. and others
Understanding Chemotherapy-Induced Peripheral Neuropathy
CIPN is a dose-dependent and often debilitating adverse effect of commonly used chemotherapeutic agents, including platinum compounds, taxanes, vinca alkaloids, and proteasome inhibitors. The condition frequently necessitates chemotherapy dose reductions or discontinuation and significantly impacts long-term quality of life in cancer survivors.
Chemotherapy-Induced Peripheral Neuropathy Epidemiology and Market Scope
In 2024, moderate CIPN cases accounted for the largest share in the U.S., with approximately 360,000 cases, followed by mild and severe forms. DelveInsight’s report analyzes epidemiological trends across the 7MM from 2020 to 2034, segmented by severity, cancer type, chemotherapeutic agent, and treatment status.
👉Support strategic planning with data-driven insights into the evolving CIPN treatment landscape
About DelveInsight
DelveInsight is a leading life sciences market research and business consulting firm, delivering end-to-end solutions to pharmaceutical and biotech companies. Through its subscription-based platform PharmDelve, DelveInsight offers seamless access to in-depth market intelligence, competitive analysis, and strategic insights to support informed decision-making and sustainable growth.