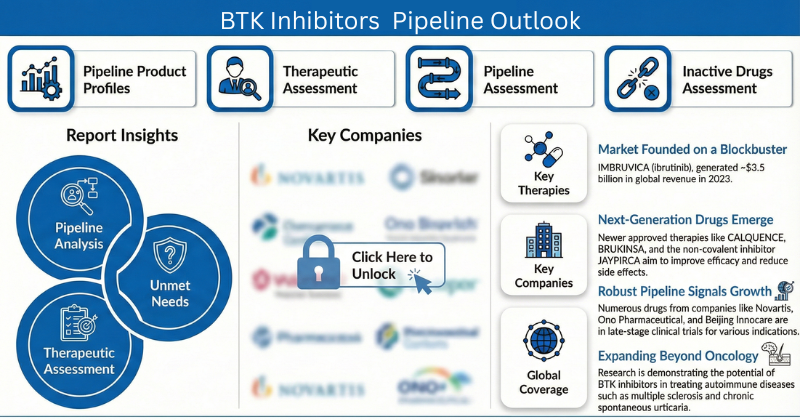
The BTK inhibitors clinical development landscape is rapidly evolving, supported by strong scientific validation and expanding therapeutic ambition. Rising incidence of B-cell malignancies, autoimmune diseases, and neurological disorders is intensifying demand for targeted therapies, positioning BTK inhibitors (BTKi) as a cornerstone of modern treatment strategies.
While first-generation BTK inhibitors have transformed outcomes in conditions such as chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), ongoing challenges including long-term safety concerns, resistance mechanisms, and high treatment costs are driving innovation toward next-generation, highly selective, and reversible BTK inhibitors. At the same time, competition from alternative targeted modalities and gene-based therapies is placing pressure on developers to clearly differentiate clinical value.
In parallel, intracerebral hemorrhage (ICH) and neuroinflammatory indications remain areas of high unmet need, where gaps in early diagnosis, disease monitoring, and effective interventions highlight both the opportunity and complexity of therapeutic advancement. Overall, the BTK inhibitors market demonstrates strong long-term potential, favoring companies that can deliver clear efficacy, improved safety profiles, and durable clinical benefits over existing standards of care.
Explore the full BTK Inhibitors clinical trial landscape BTK Inhibitors Pipeline Insight : https://www.delveinsight.com/sample-request/brutons-tyrosine-kinase-btk-inhibitor-pipeline-insight
BTK Inhibitors Pipeline Outlook
DelveInsight’s “BTK Inhibitors Pipeline Insight” provides a comprehensive global overview of pipeline therapies across all stages of clinical development. The report analyzes key trends, emerging assets, competitive positioning, and strategic developments shaping the future of the BTK inhibitors domain.
In addition, the report delivers actionable intelligence on:
- Clinical trial benchmarking
- Regulatory pathways (FDA, EMA, and global agencies)
- Licensing, partnering, and M&A activity
- Commercial and development strategies
This intelligence enables stakeholders to optimize decision-making and capture growth opportunities within the evolving BTK inhibitors landscape.
Understand competitive positioning in the BTK Inhibitors market through in-depth analysis
BTK Inhibitors Clinical Trial Landscape: Key Highlights
- 30+ pharmaceutical and biotech companies are actively developing 30+ BTK inhibitor candidates globally
- Around 2+ assets are in late-stage or registration-ready development
- 10+ BTK inhibitors are progressing through Phase I and Phase II clinical trials
- The pipeline reflects strong diversification across oncology, autoimmune, and neurological indications
Key BTK Inhibitors Companies Driving Innovation
Leading companies advancing the BTK inhibitors pipeline include:
- Lupeng Pharmaceutical
- ACEA Therapeutics
- Roche
- Merck Sharp & Dohme
- Biogen
- HUTCHMED
- Taiho Pharmaceutical
- Hanmi Pharmaceutical
- Evopoint Biosciences
- Hyperway Pharmaceutical
- HealZen Therapeutics
- InnoCare Pharma
- Crossfire Oncology
These companies are pursuing differentiated strategies focused on improved selectivity, CNS penetration, reversible binding, and expanded non-oncology indications.
Prominent BTK Inhibitors in Clinical Development
Several next-generation BTK inhibitors are showing promise across diverse indications, including:
- Rocbrutinib – R/R Mantle Cell Lymphoma
- Abivertinib – Non-Small Cell Lung Cancer
- Fenebrutinib – Multiple Sclerosis
- Nemtabrutinib – CLL/SLL
- BIIB091 – Multiple Sclerosis
- HMPL-760 – Diffuse Large B-Cell Lymphoma
- TAS5315 – Rheumatoid Arthritis
- XNW1011 (EVER001) – Primary Membranous Nephropathy
- HBW-3210 – CNS Metastases
Most candidates are oral small-molecule therapies, reflecting continued preference for patient-friendly administration and long-term use.
Identify licensing and partnering opportunities in BTK Inhibitors Clinical Trial and FDA Approval
Recent Developments in the BTK Inhibitors Space
- November 2025: Roche reported positive Phase III results for fenebrutinib in relapsing multiple sclerosis, achieving a significant reduction in annualized relapse rate versus teriflunomide
- October 2025: Zenas BioPharma secured global rights to orelabrutinib (ex-oncology), strengthening BTK inhibitors’ position in neuroinflammatory diseases
- August 2025: FDA granted Fast Track Designation to birelentinib (DZD8586) for heavily pretreated CLL/SLL patients
- May 2025: China’s CDE accepted the NDA for rocbrutinib, positioning it as a potential first domestically developed BTK inhibitor for R/R MCL
- 2024–2025: Multiple positive Phase Ib/IIa readouts and licensing deals reinforced confidence in reversible and next-generation BTK inhibitors
Scope of the BTK Inhibitors Pipeline Report
- Geographic Coverage: Global
- Therapeutic Approaches: Mono, Combination, Mono/Combination
- Clinical Stages: Discovery to Phase III
- Routes of Administration: Oral, Parenteral, Intravenous, Subcutaneous
- Molecule Types: Small molecules, monoclonal antibodies, peptides, gene-based approaches
BTK Inhibitors Strategic Outlook
The BTK inhibitors market is transitioning from first-generation oncology-focused agents toward broader, multi-indication platforms spanning immunology and neurology. Developers that successfully balance efficacy, safety, and long-term disease control are expected to define the next phase of market leadership.
Gain actionable intelligence on next-generation @ BTK Inhibitors Therapies and Drugs