(Las Vegas, Nevada, United States) According to DelveInsight’s latest evaluation, the global bispecific antibody pipeline includes more than 180 active companies collectively developing over 250 bispecific antibody–based therapeutic candidates. The pipeline analysis provides a detailed overview of clinical trials, therapeutic approaches, mechanisms of action, routes of administration, and recent development activities shaping this rapidly advancing field.
The “Bispecific Antibody Pipeline Insight, 2025” report by DelveInsight delivers an in-depth assessment of the current clinical development landscape along with future growth opportunities across the bispecific antibody market.
This comprehensive pipeline report offers a detailed clinical and commercial evaluation of bispecific antibody candidates spanning preclinical research through marketed stages. It includes extensive drug profiles covering mechanisms of action, clinical study data, regulatory milestones such as NDA approvals (where applicable), and development activities including technology platforms, strategic collaborations, mergers and acquisitions, funding updates, regulatory designations, and other key advancements.
Get a Free Sample PDF Report to know more about Bispecific Antibody Pipeline Therapeutic Assessment-
https://www.delveinsight.com/report-store/bispecific-antibody-competitive-landscape
Key Highlights from the Bispecific Antibody Pipeline Report
- Pharmaceutical and biotechnology companies worldwide are actively advancing novel bispecific antibody therapies, achieving notable clinical and developmental progress over recent years.
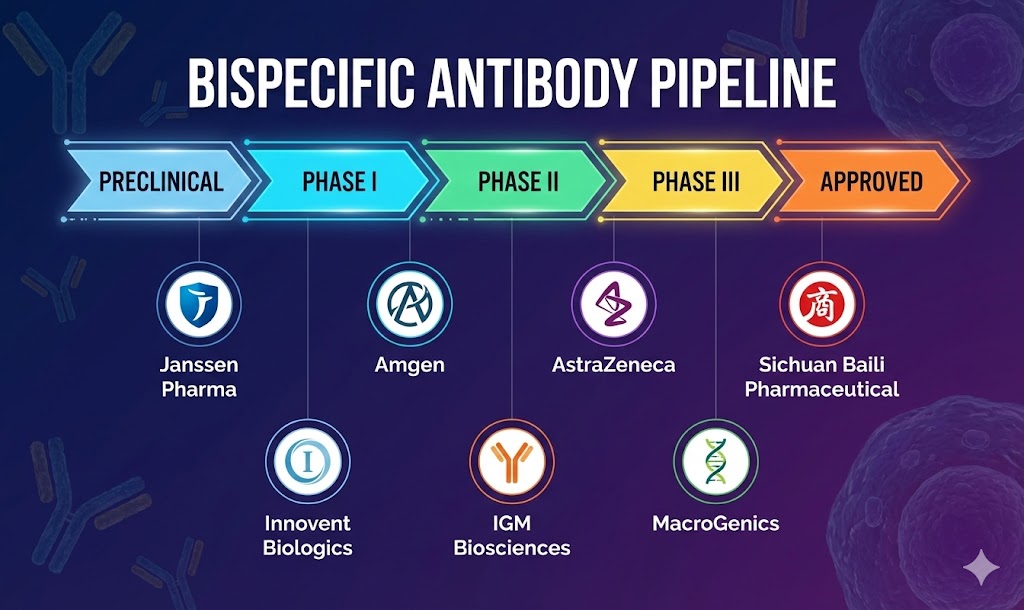
- Leading companies involved in bispecific antibody development include Janssen Pharma, Amgen, AstraZeneca, Sichuan Baili Pharmaceutical, Innovent Biologics, IGM Biosciences, MacroGenics, among others.
- Prominent bispecific antibody candidates progressing through various stages of clinical development—such as Amivantamab, Blinatumomab, Gefurulimab, SI-B001, IBI322, Imvotamab, and MGD024—are expected to significantly influence the market in the coming years.
Recent Development Updates in the Bispecific Antibody
- September 2024: Innovent Biologics received Fast Track designation from the US FDA for IBI363, a PD-1/IL-2α bispecific antibody fusion protein, for unresectable or metastatic melanoma patients previously treated with PD-1/L1 inhibitors.
- August 2024: Phanes Therapeutics secured orphan drug designation for PT217 in neuroendocrine carcinoma (NEC).
- January 2024: LAVA Therapeutics entered a clinical collaboration with Merck & Co. to evaluate LAVA-1207 in combination with KEYTRUDA® for therapy-refractory metastatic castration-resistant prostate cancer (mCRPC).
- January 2024: Biocytogen Pharmaceuticals signed an exclusive option and license agreement with Radiance Biopharma for a first-in-class anti-HER2/TROP2 bispecific ADC.
- January 2024: Debiopharm expanded its partnership with SunRock Biopharma through an exclusive licensing agreement to develop an enhanced HER3-EGFR bispecific ADC.
Further Bispecific Antibody product details are provided in the report. Download the Bispecific Antibody pipeline report to learn more about the emerging Bispecific Antibody therapies
Bispecific Antibody Overview
Bispecific antibodies are engineered immunoglobulins capable of simultaneously binding two distinct antigens or epitopes. This dual-target engagement enables immune cell redirection, enhanced immune activation, or simultaneous blockade of multiple disease pathways. Widely applied in oncology, immunology, and autoimmune disorders, bispecific antibodies offer high therapeutic precision, although their structural complexity demands advanced design and manufacturing capabilities.
Emerging Bispecific Antibody Drugs by Development Stage
- Amivantamab – Janssen Pharma
- Blinatumomab – Amgen
- Gefurulimab – AstraZeneca
- SI-B001 – Sichuan Baili Pharmaceutical
- IBI322 – Innovent Biologics
- Imvotamab – IGM Biosciences
- MGD024 – MacroGenics
Route of Administration Analysis
The report categorizes bispecific antibody candidates by route of administration, including:
- Oral
- Parenteral
- Intravenous
- Subcutaneous
- Topical
Molecule Type Classification
Bispecific antibody therapies are further segmented by molecular type, such as:
- Monoclonal antibodies
- Peptides
- Polymers
- Small molecules
- Gene therapies
Pipeline Therapeutic Assessment Framework
- Assessment by product type and development stage
- Route of administration analysis by stage
- Molecule type evaluation across clinical phases
DelveInsight’s report evaluates more than 250 bispecific antibody candidates across:
- Late-stage (Phase III)
- Mid-stage (Phase II)
- Early-stage (Phase I)
- Preclinical and discovery programs
- Discontinued or inactive assets
Bispecific Antibody Competitive Landscape
Major companies active in the bispecific antibody therapeutics market include Roche, Amgen, Regeneron Pharmaceuticals, AstraZeneca, Novartis, Merck & Co., Pfizer, Eli Lilly, and others.
Download Sample PDF Report to know more about Bispecific Antibody drugs and therapies
Bispecific Antibody Pipeline Market Drivers
- Increasing demand for targeted and precision therapies
- Strong clinical success in oncology indications
- Advancements in antibody engineering technologies
- Rising R&D investments and strategic partnerships
- Regulatory incentives such as orphan drug and fast-track designations
Bispecific Antibody Pipeline Market Barriers
- Complex design and manufacturing requirements
- Safety concerns including cytokine release syndrome
- High development and commercialization costs
- Regulatory and clinical trial complexity
- Competition from alternative treatment modalities such as CAR-T therapies and ADCs
Scope of the Bispecific Antibody Pipeline Insight
- Geographical Coverage: Global
- Key Companies: Janssen Pharma, Amgen, AstraZeneca, Sichuan Baili Pharmaceutical, Innovent Biologics, IGM Biosciences, MacroGenics, and others
- Key Therapies: Amivantamab, Blinatumomab, Gefurulimab, SI-B001, IBI322, Imvotamab, MGD024, and others
- Therapeutic Assessment: Marketed and emerging bispecific antibody therapies
- Market Dynamics: Drivers and barriers influencing pipeline growth
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance. It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate business growth and overcome challenges with a practical approach.