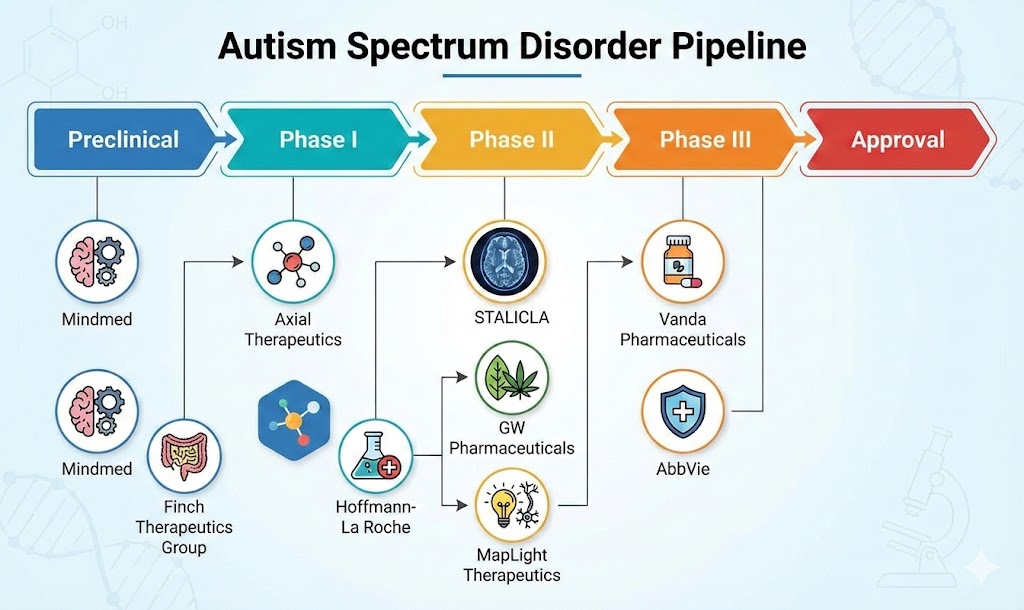
The Autism Spectrum Disorder (ASD) Pipeline Insight 2025 highlights a rapidly evolving therapeutic landscape, driven by increasing disease prevalence, advances in neurodevelopmental research, and growing investment in precision medicine. According to DelveInsight, more than 18 biopharmaceutical companies are actively developing 20+ investigational therapies for Autism Spectrum Disorder across preclinical, Phase I, Phase II, and Phase III clinical trials.
The Autism Spectrum Disorder Pipeline Report 2025 provides comprehensive coverage of clinical trial milestones, mechanisms of action, routes of administration, molecular targets, and regulatory designations, offering critical insights into the future of ASD treatment.
Get a Free Sample PDF Report to know more about Autism Spectrum Disorder Pipeline Therapeutic Assessment-
https://www.delveinsight.com/report-store/autism-spectrum-disorder-asd-pipeline-insight
Autism Spectrum Disorder Drug Development Landscape
The Autism Spectrum Disorder pipeline spans a broad range of therapeutic strategies, including small molecules, biologics, gene therapies, cannabinoid-based treatments, and microbiome-modulating therapies. These innovative approaches aim to address core ASD symptoms, such as social communication deficits, behavioral rigidity, sensory sensitivity, and cognitive impairment.
Leading companies advancing Autism Spectrum Disorder therapeutics include Mindmed, Finch Therapeutics Group, Axial Therapeutics, Hoffmann-La Roche, STALICLA, GW Pharmaceuticals, MapLight Therapeutics, Vanda Pharmaceuticals, AbbVie, Novartis, and Otsuka Pharmaceutical, among others.
Emerging Autism Spectrum Disorder Therapies
Several pipeline candidates are expected to shape the future Autism Spectrum Disorder treatment paradigm, including:
- MM-402 (Mindmed)
- FIN 211 (Finch Therapeutics Group)
- AB-2004 / AB-2004 PTR (Axial Therapeutics)
- RO6953958 (Hoffmann-La Roche)
- STP-1 (STALICLA)
- Cannabidiol (GW Pharmaceuticals)
- ML-004 (MapLight Therapeutics)
- Tasimelteon (Vanda Pharmaceuticals)
- Cariprazine (AbbVie)
These candidates target diverse biological pathways such as neurotransmitter modulation, gut-brain axis signaling, circadian rhythm regulation, synaptic function, and neuroinflammation.
Further Autism Spectrum Disorder product details are provided in the report. Download the Autism Spectrum Disorder pipeline report to learn more about the emerging Autism Spectrum Disorder therapies
Recent Clinical and Regulatory Developments
- In May 2025, Yamo Pharmaceuticals reported positive Phase II trial results for L1-79, demonstrating statistically significant improvements in socialization outcomes in adolescents and young adults with ASD.
- In February 2025, the FDA cleared DeFloria’s IND, allowing Phase II development of AJA001 Oral Solution, a botanical drug for ASD symptoms.
- In January 2025, SciSparc’s SCI-210 clinical trial for pediatric ASD patients received renewal approval from Israel’s Medical Cannabis Agency.
- In July 2024, Jaguar Gene Therapy received FDA feedback supporting the advancement of JAG201, a gene replacement therapy for SHANK3-related autism and Phelan-McDermid syndrome.
Autism Spectrum Disorder Market Drivers and Barriers
Key Autism Spectrum Disorder Market Drivers
- Rising global Autism Spectrum Disorder prevalence rates
- Growing demand for early diagnosis and targeted therapies
- Increased R&D investment in neurodevelopmental disorders
- Regulatory incentives for orphan and pediatric drug development
Key Autism Spectrum Disorder Market Barriers
- Limited FDA-approved pharmacologic treatments
- High treatment and diagnostic costs
- Clinical heterogeneity and complex trial endpoints
- Long development timelines and regulatory challenges
Download Sample PDF Report to know more about Autism Spectrum Disorder drugs and therapies
Conclusion:
The Autism Spectrum Disorder Pipeline 2025 reflects accelerating innovation across precision therapeutics, gene therapy, microbiome science, and neuropharmacology. While challenges remain, the expanding pipeline signals meaningful progress toward disease-modifying and symptom-targeted treatments that could significantly improve quality of life for individuals with Autism Spectrum Disorder.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing end-to-end comprehensive solutions to improve their performance.
Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve.