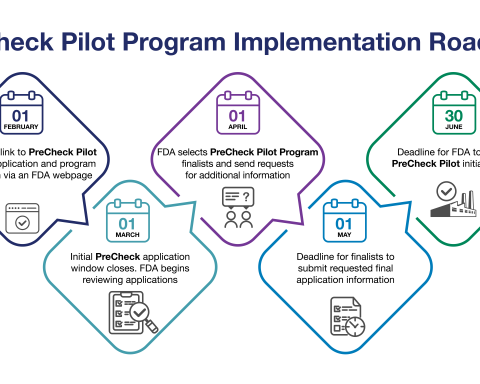
FDA Launches PreCheck Pilot Program to Accelerate U.S. Pharmaceutical Manufacturing; Applications Open February 1, 2026
The U.S. Food and Drug Administration announced that it will begin accepting applications on February 1, 2026, for the FDA PreCheck Pilot Program, a transformative initiative designed to strengthen the domestic pharmaceutical supply chain by increasing regulatory predictability and accelerating the development

