DelveInsight’s “Lassa Fever – Pipeline Insight, 2026” provides a detailed view of the evolving pipeline, emphasizing efforts to deliver first-in-class therapies and vaccines to endemic populations. The report highlights global R&D activity, platform technologies, and regulatory incentives accelerating development timelines.
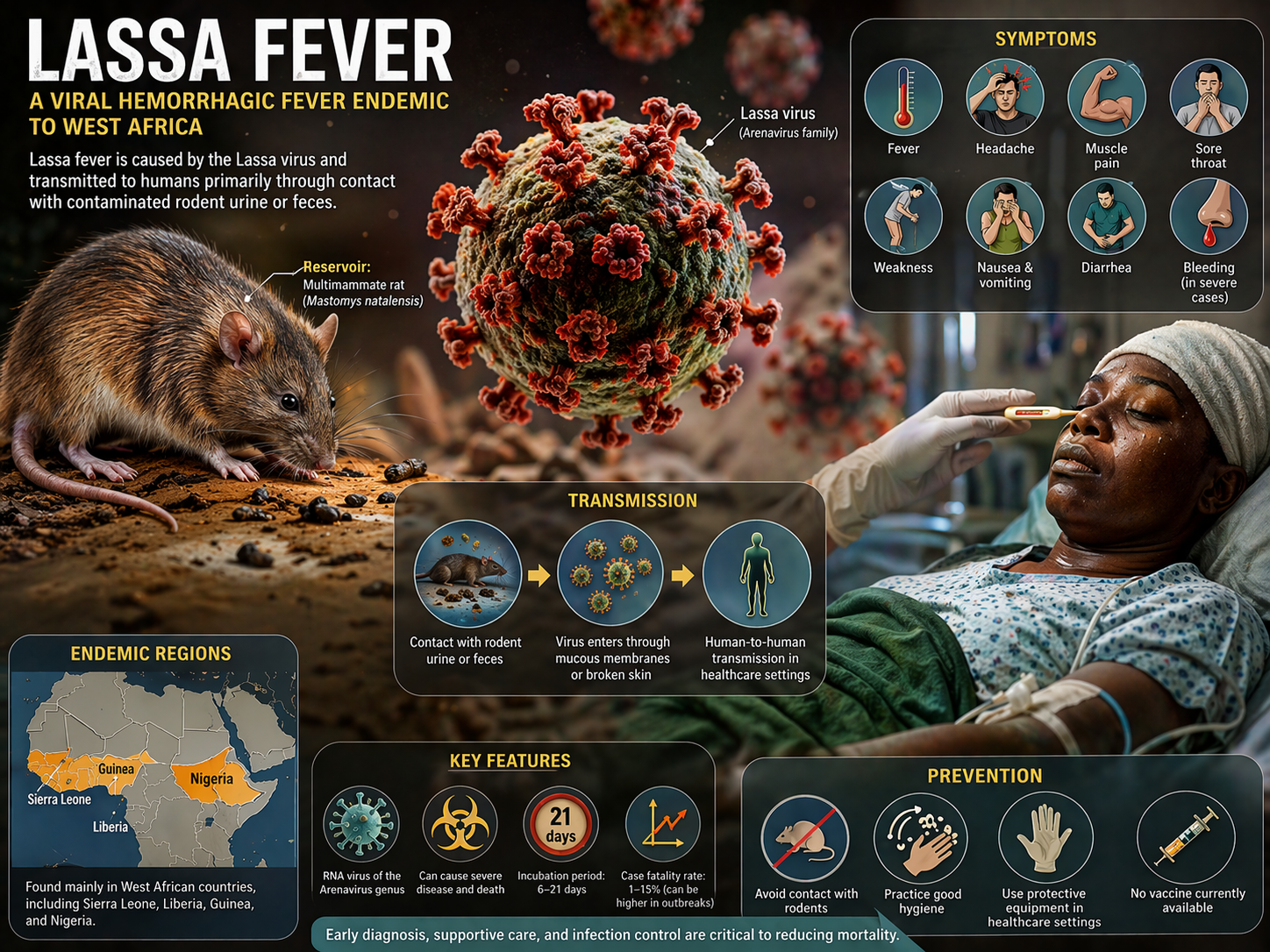
Lassa Fever, an acute viral hemorrhagic disease endemic to West Africa, remains a serious public health challenge due to recurrent outbreaks and limited treatment options. Despite high fatality rates in severe cases, management is still mainly supportive, highlighting the urgent demand for effective antivirals and preventive vaccines.
By 2025, the Lassa fever pipeline includes over 3 active candidates, such as direct-acting antivirals, monoclonal antibodies, and next-generation vaccines. Leading players like Arisan Therapeutics and Imunon are advancing programs supported by international collaborations and expedited regulatory frameworks.
With increased focus on global pandemic preparedness, the Lassa fever pipeline is well-positioned to transform outbreak response, bringing scalable and sustainable solutions to endemic regions and beyond.
Interested in learning more about the current treatment landscape and the key drivers shaping the Lassa Fever pipeline? Click here: https://www.delveinsight.com/report-store/lassa-fever-pipeline-insight
Key Takeaways from the Lassa Fever Pipeline Report
- DelveInsight’s pipeline review highlights 3+ active companies developing 3+ pipeline drugs for Lassa Fever.
- Leading companies such as Arisan Therapeutics and Imunon are advancing assets aimed at improving Lassa Fever management.
- Prominent candidates include ARN-75039, IMNN-102, and others.
- March 2025: Institut Pasteur, Paris, launched a Phase Ia trial of MOPEVAC-Lassa, a viral vector vaccine, enrolling 72 volunteers with dosing planned for 2026.
- March 2025: The US NIH/NIAID initiated a Phase I trial for LASSARAB, a rabies-vectored vaccine, enrolling up to 55 adults at the University of Maryland.
- January 2025: The West Africa Lassa Fever Consortium (WALC) introduced a multi-country Phase II/III adaptive trial framework to evaluate multiple therapies in endemic regions.
- August 2024: IAVI began a multisite Phase II trial in Ghana, Liberia, and Nigeria for its Lassa vaccine, enrolling 612 participants (adults, adolescents, and children).
Request a sample and discover the recent breakthroughs happening in the Lassa Fever pipeline landscape at : https://www.delveinsight.com/report-store/lassa-fever-pipeline-insight
Lassa Fever Overview
Lassa fever is a viral hemorrhagic illness caused by the Lassa virus of the Arenaviridae family. Endemic in West Africa (notably Nigeria, Sierra Leone, Liberia, and Guinea), it is spread mainly through contact with food or household items contaminated by infected Mastomys rats. Human-to-human transmission also occurs, especially in healthcare settings lacking infection control.
While most infections are mild or asymptomatic, severe cases can lead to multi-organ failure, hemorrhage, neurological issues, respiratory distress, and death. The incubation period is 6–21 days, with symptoms often mimicking other febrile diseases like malaria, complicating diagnosis.
There is currently no approved vaccine. Treatment is mainly supportive, though ribavirin has shown some benefit when given early. Due to its severity, outbreak potential, and risk to healthcare workers, Lassa fever is designated by the WHO as a priority pathogen, spurring global efforts to develop vaccines, monoclonal antibodies, and antivirals.
Find out more about Lassa Fever medication at: https://www.delveinsight.com/report-store/lassa-fever-pipeline-insight
Lassa Fever Treatment Analysis: Drug Profile
ARN-75039 (Arisan Therapeutics)
ARN-75039 is a broad-spectrum entry inhibitor targeting arenaviruses. Preclinical studies in guinea pigs demonstrated strong antiviral activity with oral dosing (60 mg/kg for 14 days), reducing severity and improving survival. Currently in Phase I clinical trials, ARN-75039 represents a key step toward advancing therapies for Lassa Fever and related viral hemorrhagic infections.
Learn more about the novel and emerging Lassa Fever pipeline therapies at: https://www.delveinsight.com/report-store/lassa-fever-pipeline-insight
Lassa Fever Therapeutics Assessment
By Product Type
- Mono
- Combination
- Mono/Combination
By Stage
- Late-stage products (Phase III)
- Mid-stage products (Phase II)
- Early-stage products (Phase I)
- Pre-clinical and Discovery candidates
- Discontinued & Inactive candidates
By Route of Administration
- Oral
- Parenteral
- Intravenous
- Subcutaneous
- Topical
By Molecule Type
- Recombinant fusion proteins
- Small molecules
- Monoclonal antibodies
- Peptides
- Polymers
- Gene therapies
Dive deep into rich insights for drugs used for Lassa Fever treatment, visit: https://www.delveinsight.com/report-store/lassa-fever-pipeline-insight
Scope of the Lassa Fever Pipeline Report
- Coverage: Global
- Key Companies: Arisan Therapeutics, Imunon, and others
- Key Pipeline Therapies: ARN-75039, IMNN-102, and others
- Therapeutic Assessment by Product Type: Mono, Combination, Mono/Combination
- Therapeutic Assessment by Clinical Stage: Discovery, Pre-clinical, Phase I, Phase II, Phase III