DelveInsight’s “Pulmonary Hypertension associated with Interstitial Lung Disease Market Insights, Epidemiology, and Market Forecast – 2034” report delivers an in-depth understanding of Pulmonary Hypertension associated with Interstitial Lung Disease, historical and forecasted epidemiology, as well as the Pulmonary Hypertension associated with Interstitial Lung Disease market trends in the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan.
Discover Key Insights into the Pulmonary Hypertension associated with Interstitial Lung Disease Market with DelveInsight’s In-Depth Report @ https://www.delveinsight.com/sample-request/pulmonary-hypertension-associated-with-interstitial-lung-disease-ph-ild-market
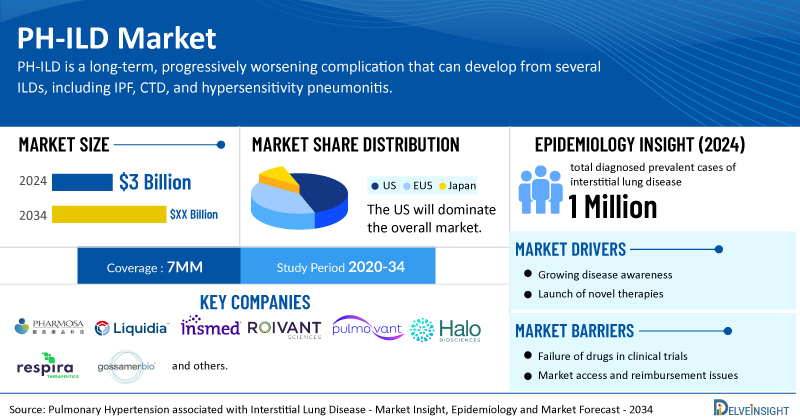
Key Takeaways from the Pulmonary Hypertension associated with Interstitial Lung Disease Market Report
- In January 2026- Pulmovant Inc. announced a Phase 2, open-label, multi-center clinical study to evaluate the safety and efficacy of inhaled mosliciguat in participants with pulmonary hypertension associated with interstitial lung disease (PH-ILD) on a background inhaled treprostinil.
- In January 2026- GB002 Inc. conducted a Phase 3 study is designed as a 24-week randomized, double-blind, placebo-controlled period (PCP) followed by a 144-week long-term extension (LTE) period. The primary objective of the PCP is to evaluate the effect of seralutinib on improving exercise capacity in subjects with World Health Organization (WHO) Group 3 pulmonary hypertension associated with interstitial lung disease. The primary objective of the LTE is to evaluate the long-term safety and tolerability of seralutinib.
- In January 2026, Insmed Incorporated initiated a phase 3, Randomized, Double-blind, Placebo-controlled, Multicenter, Parallel Group Study to Evaluate the Efficacy and Safety of Treprostinil Palmitil Inhalation Powder in Participants With Pulmonary Hypertension Associated With Interstitial Lung Disease.
- In September 2025, AllRock Bio announced a $50 million Series A funding round, co-led by Versant Ventures and Westlake BioPartners, to advance ROC-101 into Phase 2 clinical development. ROC-101 is a first-in-class oral pan-ROCK inhibitor targeting pulmonary arterial hypertension (PAH) and pulmonary hypertension with interstitial lung disease (ILD-PH), addressing critical unmet needs in these life-threatening conditions by modulating inflammatory, proliferative, and fibrotic remodeling.
- In 2024, in the US, diagnosed prevalence of PH-ILD showed a slightly higher burden in males, with approximately 42,000 cases compared to 39,000 in females. This gender gap is expected to widen by 2034, highlighting potential differences in disease susceptibility or progression.
- In 2024, an estimated 69,000 people in the EU4 and the UK were diagnosed with PH-ILD, underscoring a substantial and growing regional disease burden.
- In France, CTD linked with PH-ILD had the highest diagnosed prevalence, with nearly 4,000 cases in 2024, while hypersensitivity pneumonitis reported the lowest burden, with just over 500 cases.
- In Japan, over 28,000 diagnosed prevalent cases of PH-ILD were reported in 2024, with the numbers projected to increase by 2034, indicating a rising disease burden.
- In 2024, IPF accounted for the largest share of PH-ILD cases in Japan at 21%, while hypersensitivity pneumonitis represented just 1%, underscoring notable variation in subtype distribution and the need for tailored diagnostic and treatment strategies.
- The leading Pulmonary Hypertension associated with Interstitial Lung Disease Companies such as Pharmosa BioPharm, Liquidia Corporation, Insmed, Roivant Sciences Pulmovant and others.
- Promising Pulmonary Hypertension associated with Interstitial Lung Disease Therapies such as Treprostinil Palmitil Inhalation Powder, Riociguat (Adempas, BAY63-2521), Mosliciguat, Mirivadelgat, and others.
Stay ahead in the Pulmonary Hypertension associated with Interstitial Lung Disease Therapeutics Market with DelveInsight’s Strategic Report @ Pulmonary Hypertension associated with Interstitial Lung Disease Market Outlook
Pulmonary Hypertension associated with Interstitial Lung Disease Epidemiology Segmentation in the 7MM
- Pulmonary Hypertension associated with Interstitial Lung Disease Diagnosed prevalent cases
- Pulmonary Hypertension associated with Interstitial Lung Disease Type-specific Diagnosed Prevalent Cases
- Pulmonary Hypertension associated with Interstitial Lung Disease Type-specific Diagnosed Prevalent Cases
- Pulmonary Hypertension associated with Interstitial Lung Disease Diagnosed Prevalent Cases
Download the report to understand which factors are driving Pulmonary Hypertension associated with Interstitial Lung Disease epidemiology trends @ Pulmonary Hypertension associated with Interstitial Lung Disease Prevalence
Pulmonary Hypertension associated with Interstitial Lung Disease Marketed Drugs
- YUTREPIA (Treprostinil): Liquidia Corporation
YUTREPIA is a dry powder inhalation formulation of treprostinil sodium, a prostacyclin mimetic designed for the treatment of PAH. Delivered through a single-use, handheld inhaler, each capsule contains treprostinil sodium blended with carefully selected excipients L-leucine, polysorbate 80, sodium chloride, sodium citrate, and trehalose.
- TYVASO/TYVASO DPI/TREPROST (treprostinil): United Therapeutics
TYVASO and TYVASO DPI (treprostinil) are approved inhaled therapies for PAH (Group 1) and PH-ILD (Group 3), helping improve exercise capacity in adults, though effects may diminish after ~4 hours. In Japan, treprostinil is available as TREPROST, but it has been withdrawn from European markets.
Pulmonary Hypertension associated with Interstitial Lung Disease Emerging Drugs
- Treprostinil liposomal (L606): Pharmosa Biopharm/Liquidia Corporation
L606 is a liposomal formulation of treprostinil, with liposome particle sizes ranging from 110–140 nm, suspended in a sodium citrate–bicarbonate buffer. This advanced formulation is engineered to enhance drug stability and prolong systemic circulation by protecting treprostinil from rapid degradation and clearance. The liposomal encapsulation enables a sustained pulmonary release, allowing for more consistent drug exposure over time, while minimizing peaks and troughs that may lead to side effects. The citrate–bicarbonate buffer not only helps maintain an optimal pH for lung tolerability but also supports better drug solubility and absorption.
- Treprostinil Palmitil Inhalation Powder (TPIP): Insmed
TPIP is a next-generation, once-daily dry powder formulation of a treprostinil prodrug, delivered via a convenient capsule-based inhaler. Engineered by Insmed, TPIP features a 16-carbon ester-linked modification for extended activity and is being developed for PAH, PH-ILD, and other severe pulmonary conditions. With promising Phase IIb results, a pivotal Phase III trial is expected to launch in the second half of 2025, marking a major step toward transforming treatment for rare lung diseases.
- Mosliciguat: Roivant Sciences/Pulmovant
Mosliciguat (BAY 1237592) is a first-in-class sGC activator being developed for PH-ILD, offering a nitric oxide–independent mechanism that promotes vasodilation even under hypoxic and oxidative stress conditions. It may also provide anti-inflammatory and anti-fibrotic benefits. Phase II topline results are expected in H2 2026.
Get In-Depth Knowledge on Pulmonary Hypertension associated with Interstitial Lung Disease Market Trends and Forecasts with DelveInsight @ Pulmonary Hypertension associated with Interstitial Lung Disease Treatment Market
Pulmonary Hypertension associated with Interstitial Lung Disease Treatment Market
The PH-ILD treatment landscape is shifting from supportive care to targeted, disease-modifying approaches that address vascular remodeling, fibrosis, and inflammation. Traditional strategies, including ILD management and lung transplantation, offer limited impact on long-term outcomes, highlighting the need for more effective therapies. New drug classes with lung-targeted mechanisms are advancing PH-ILD treatment. Inhaled prostacyclins like treprostinil have enhanced exercise capacity, setting the stage for next-generation agents such as Mosliciguat (sGC activator), H1614 (HA synthesis inhibitor), and RT234 (inhaled PDE5 inhibitor). These candidates aim to deliver broader benefits—beyond vasodilation—by incorporating anti-fibrotic and anti-inflammatory effects, marking a shift toward disease-modifying approaches.
Pulmonary Hypertension associated with Interstitial Lung Disease Market Outlook
The market for PH-ILD is undergoing significant transformation, driven by growing disease recognition, evolving clinical strategies, and novel therapeutic development. PH-ILD is the most common form of Group 3 pulmonary hypertension and represents a serious complication of ILD, contributing to cardiopulmonary disability, right heart dysfunction, and increased mortality. Despite supportive treatments such as oxygen therapy and corticosteroids to manage acute exacerbations, current standards offer limited long-term benefit and do not halt disease progression.
Unlock Strategic Insights with DelveInsight’s Comprehensive Pulmonary Hypertension associated with Interstitial Lung Disease Market Report @ Pulmonary Hypertension associated with Interstitial Lung Disease Market Drivers and Barriers
Scope of the Pulmonary Hypertension associated with Interstitial Lung Disease Market Report
- Coverage- 7MM
- Study Period- 2020-2034
- Pulmonary Hypertension associated with Interstitial Lung Disease Companies- Pharmosa BioPharm, Liquidia Corporation, Insmed, Roivant Sciences Pulmovant and others.
- Pulmonary Hypertension associated with Interstitial Lung Disease Therapies- Treprostinil Palmitil Inhalation Powder, Riociguat (Adempas, BAY63-2521), Mosliciguat, Mirivadelgat, and others.
- Pulmonary Hypertension associated with Interstitial Lung Disease Competitive Intelligence Analysis: SWOT analysis and Market entry strategies
- Pulmonary Hypertension associated with Interstitial Lung Disease Unmet Needs, KOL’s views, Analyst’s views, Pulmonary Hypertension associated with Interstitial Lung Disease Market Access and Reimbursement
Table of Content
1: Key Insights
2: PH-ILD Market Report Introduction
3: Pulmonary Hypertension Associated with Interstitial Lung Disease Market Overview at a Glance
4: PH-ILD Epidemiology and Market Methodology
5: PH-ILD Executive Summary
6: Key Events
7: PH-ILD Disease Background and Overview
8: PH-ILD Epidemiology and Patient Population
9: PH-ILD Patient Journey
10: PH-ILD Marketed drugs
11: PH-ILD Emerging Drugs
12. Key Cross Competition
13: PH-ILD Unmet Needs
14: PH-ILD SWOT Analysis
15: Key Opinion Leaders’ Views
16: Reimbursement and PH-ILD Market Access
17: Appendix
18: DelveInsight Capabilities
19: Disclaimer
20: About DelveInsight
About Us
DelveInsight is a leading healthcare-focused market research and consulting firm that provides clients with high-quality market intelligence and analysis to support informed business decisions. With a team of experienced industry experts and a deep understanding of the life sciences and healthcare sectors, we offer customized research solutions and insights to clients across the globe. Connect with us to get high-quality, accurate, and real-time intelligence to stay ahead of the growth curve.