The chronic refractory cough (CRC) market is projected to witness substantial growth through 2034, driven by an expanding diagnosed patient population, increasing recognition of chronic cough as a distinct clinical condition, and the shortcomings of currently used off-label therapies.
Chronic Refractory Cough Market momentum is further supported by the anticipated launch of innovative therapies with novel mechanisms of action, including camlipixant (GSK), Haduvio (nalbuphine ER; Trevi Therapeutics), and Taplucainium (Nocion Therapeutics), which aim to address the significant unmet needs in refractory patient populations.
👉Unlock comprehensive market forecasts, epidemiology trends, and pipeline analysis for Chronic Refractory Cough through 2034: https://www.delveinsight.com/sample-request/chronic-refractory-cough-market
Chronic Refractory Cough Market Outlook
According to DelveInsight’s Chronic Refractory Cough Market Insights report, the market encompasses a detailed analysis of current treatment paradigms, emerging therapeutic candidates, individual drug market shares, and market size forecasts spanning 2020 to 2034 across major geographies, including the United States, EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
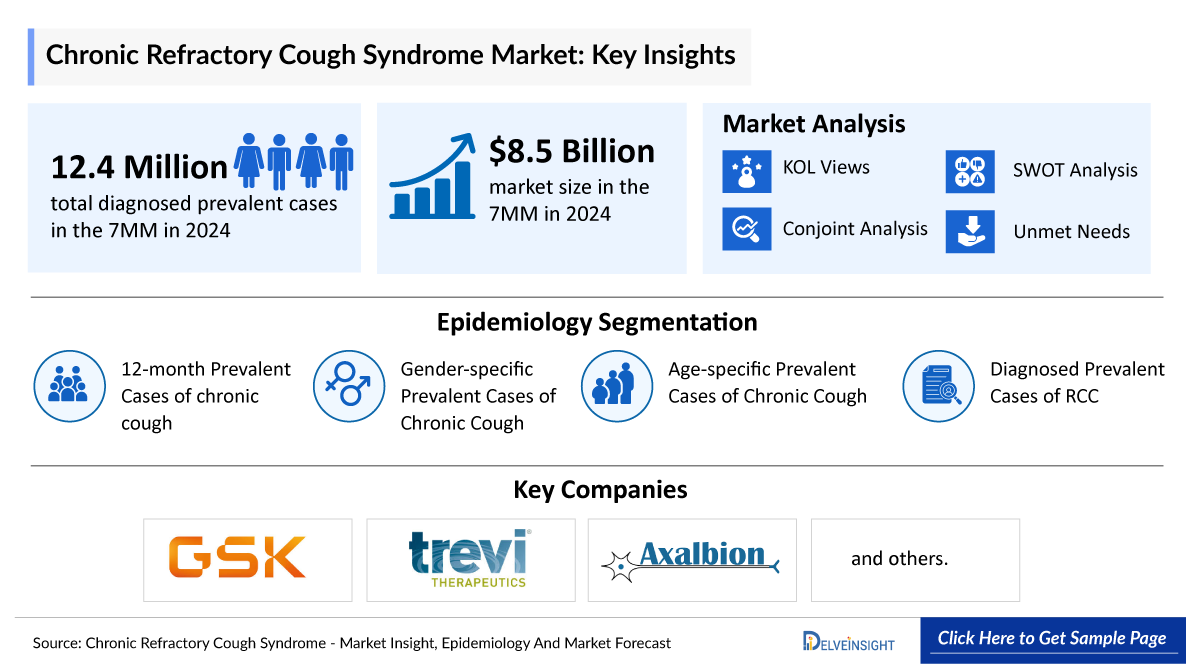
In 2024, the total chronic refractory cough market across these leading regions was valued at approximately USD 9 billion. The United States dominated the landscape, accounting for nearly 50% of the total market share within the 7MM, outperforming Europe and Japan.
During the same year, the estimated 12-month prevalent chronic cough population in the 7MM reached nearly 31 million, with the U.S. contributing close to 13 million cases. Pharmaceutical innovators such as GSK, Trevi Therapeutics, and Nocion Therapeutics are actively advancing novel CRC therapies, many of which are expected to enter the market during the forecast period and reshape the treatment paradigm.
Key Growth Drivers of the Chronic Refractory Cough Market
Rising Disease Prevalence
The growing burden of chronic cough, particularly among aging populations, continues to expand the addressable market.
Increased susceptibility to comorbidities such as asthma, chronic obstructive pulmonary disease (COPD), and other respiratory disorders is expected to further elevate CRC prevalence through 2034.
Advancements in Diagnostic Capabilities
The adoption of advanced diagnostic tools including cough reflex sensitivity assessments, high-resolution computed tomography (HRCT), and refined clinical algorithms has significantly improved diagnostic accuracy. Enhanced detection supports personalized treatment strategies and facilitates greater uptake of specialized therapies.
Emergence of Novel Therapies
The anticipated approval and commercialization of next-generation therapies such as camlipixant, Haduvio, and Taplucainium are expected to be key catalysts for market growth during the 2025–2034 forecast period.
👉Understand market size, growth drivers, and competitive dynamics shaping the Chronic Refractory Cough market
Chronic Refractory Cough Treatment Landscape
Management of chronic refractory cough typically requires a multidisciplinary approach, combining pharmacologic and non-pharmacologic interventions.
Current pharmacologic strategies focus on mitigating neural hypersensitivity and often include neuromodulators, proton pump inhibitors (PPIs) for reflux-associated symptoms, inhaled corticosteroids (ICS), and antitussives.
Non-pharmacologic approaches such as speech therapy, cough suppression techniques, cognitive behavioral therapy, and airway clearance methods aim to improve symptom control and quality of life.
Notably, there is no FDA-approved therapy for CRC in the United States at present. Treatment primarily relies on off-label agents including opioids (morphine, codeine, tramadol), gabapentin, pregabalin, amitriptyline, and baclofen.
In contrast, LYFNUA (gefapixant) has gained regulatory approval in Japan (2022) and the European Union (2023) for adults with refractory or unexplained chronic cough, highlighting growing global recognition of targeted CRC therapies.
Chronic Refractory Cough Pipeline Developments
The CRC therapeutic pipeline is rapidly evolving, with several late-stage candidates poised to transform the market:
- Camlipixant (GSK): A highly selective oral P2X3 receptor antagonist currently undergoing evaluation in two pivotal Phase III trials (CALM-I and CALM-II). Topline results are expected in H2 2025, with regulatory submissions anticipated in 2026.
- Haduvio (nalbuphine ER; Trevi Therapeutics): A dual-acting agent functioning as a kappa-opioid receptor agonist and µ-opioid receptor antagonist. Its mechanism enables cough suppression through both central and peripheral pathways and is being explored in chronic cough associated with idiopathic pulmonary fibrosis (IPF) and renal cell carcinoma.
- Taplucainium (Nocion Therapeutics): A novel inhaled charged sodium channel blocker designed to selectively silence activated pulmonary nociceptors while minimizing systemic exposure. The ongoing Phase IIb ASPIRE trial is assessing its efficacy as a once-daily inhaled therapy in refractory or unexplained chronic cough.
The successful launch of these agents is expected to redefine standards of care, intensify competition, and unlock new commercial opportunities within the CRC market.
Understanding Chronic Refractory Cough
Chronic refractory cough is defined as a cough persisting for more than eight weeks that remains unresponsive to standard treatments, even after thorough evaluation and management of common underlying causes such as asthma, GERD, or postnasal drip.
Often associated with cough hypersensitivity syndrome, CRC significantly impairs quality of life, leading to sleep disturbances, physical exhaustion, social limitations, and psychological distress.
👉Strengthen your commercial strategy with DelveInsight’s deep-dive analysis of the Chronic Refractory Cough treatment market
Chronic Refractory Cough Competitive Landscape
The chronic refractory cough competitive landscape is rapidly evolving as the unmet need for targeted therapies draws intense R&D focus. Currently, no U.S.-approved treatments exist, and management relies on off-label neuromodulators (e.g., gabapentin) and non-pharmacologic care.
Emerging frontrunners are P2X3 receptor antagonists like gefapixant (Lyfnua), approved in the EU and Japan, and camlipixant (GSK) in Phase III with expected regulatory filings. Other innovative candidates include Haduvio (nalbuphine ER) and Taplucainium, highlighting diversification beyond P2X3 mechanisms.
Major pharma (Merck, GSK, Shionogi) and smaller biotechs are investing in this space, signaling a likely competitive first-wave of approvals.
Key Chronic Refractory Cough Market Companies
- GSK,
- Trevi Therapeutics,
- Nocion Therapeutics,
- Merck,
- Kyorin Pharmaceuticals, among others
Chronic Refractory Cough Epidemiology Insights
In 2024, gender-specific analysis in the United States revealed approximately 5.1 million male and 8 million female chronic cough cases. Higher prevalence among females is attributed to anatomical airway differences and increased sensitivity to environmental triggers.
DelveInsight’s epidemiology assessment spans 2020–2034 and includes:
- 12-month prevalent chronic cough cases
- Gender-specific prevalence
- Diagnosed chronic refractory cough cases
- Chronic cough prevalence in idiopathic pulmonary fibrosis
Chronic Refractory Cough Market Report Highlights
- Study Period: 2020–2034
- Geographic Coverage: 7MM (US, EU4, UK, Japan)
- Market Size (2024): USD 9 billion
- Key Companies: GSK, Trevi Therapeutics, Nocion Therapeutics, Merck, Kyorin Pharmaceuticals, among others
- Leading Therapies: Camlipixant, Haduvio, Taplucainium, LYFNUA
The report further provides in-depth analysis of market dynamics, unmet needs, competitive intelligence, access and reimbursement trends, and insights from key opinion leaders (KOLs).
👉Connect with our experts to gain bespoke insights into Chronic Refractory Cough market opportunities